The Hard Challenge: Making Implants Soft and Smart

Most tissues in the body are soft. Yet, when doctors want to strengthen a weak spot in an artery or keep a clogged artery open, they use hard plastic or metal tubes called stents. Sometimes, the hard, inflexible materials cause problems. That is why scientists from the University of Groningen and the University Medical Center Groningen are developing stents that are made of softer materials. They aim to do something similar for valves that are implanted into the lungs to treat patients with severe emphysema.
FSE Science Newsroom / René Fransen
‘The materials that implants are made of often cause inflammation or tissue damage,’ explains Marleen Kamperman, Professor of Polymer Science at the Faculty of Science and Engineering (FSE). ‘So, the first step is to design soft materials that could replace them. And we are also looking for less invasive means to insert such implants.’
Tuning stiffness
Kamperman leads the project together with Patrick van Rijn, Professor of Translational and Regenerative Biomaterials at the University Medical Center Groningen (UMCG). ‘We want to redesign these hard implants,’ he says. An interesting alternative is the use of hydrogels: polymers that can absorb large amounts of water. These gels can be made harder or softer, and their stiffness can be tuned to that of surrounding tissues.
Another advantage of the hydrogels is that they shrink when dehydrated. This makes it easier to insert them into the body, for example via injection. Once in place, gels can be induced to absorb water and expand. ‘And there are ways to make the material stiffer after placement,’ says Van Rijn.
And there is more room for improvement. To insert stents or valves, they are pushed through a major blood vessel or the bronchi of the lungs using a plastic catheter. This is a rather invasive method. By adding magnetic materials to the hydrogel, the implant could be transported through blood vessels with the help of a magnetic field.
PhD student Adriana Vasi is working out a way to guide a stent to its destination using magnets and has experienced first-hand how a smaller, shrunken version is much easier to manoeuvre through the vessel than a full-sized stent that is made of a hard material (see video and text box below).
Ambitious innovations
This research project ‘Minimally Invasive Implants Using Smart Soft Materials’ started with a large grant from the Ubbo Emmius Foundation for HTRIC, the Health Technology Research and Innovation Cluster of the University of Groningen and the UMCG. The strength of HTRIC lies in the combination of materials science expertise from the FSE and clinical knowledge and experience from the UMCG.
Minimally Invasive Implants Using Smart Soft Materials
In 2025, the University of Groningen’s Ubbo Emmius Fund awarded HTRIC a grant of €18.4 million. This grant supports innovative research into health technology and strengthens the Northern Netherlands as a key region in this field. The research programme focuses on four innovative themes: molecular diagnostics, imaging, targeted therapies, and new materials. Research into soft implants falls under the latter theme.
Principal scientists:
Prof. Dr. Patrick van Rijn (UMCG) Professor Materiobiology & Nanobiomaterials
Dr. Gosia Wlodarczyk-Biegun (FSE) Assistant Professor Biofabrication and Bio-Instructive Materials
Prof. Dr. Marleen Kamperman (FSE) Professor of Polymer Science
Dr. Michael Lerch (FSE) Assistant Professor Autonomous Soft Materials
Dr. Andrea Giuntoli (FSE) Assistant Professor of Micromechanics Computational Modeling
Prof. Dr. Antonis Vakis (FSE) Assistant Professor of Computational Mechanics and Materials Engineering
Prof. Dr. Jean-Paul de Vries (UMCG) Professor of Cardiovascular Surgery, Head of Department of Surgery
Prof. Dr. Dirk-Jan Slebos (UMCG) Professor of Pulmonology, Head of the Department of Pulmonology and Tuberculosis
Prof. Dr. Ajay Giri Prakash Kottapalli (FSE) Associate Professor of Bioinspired MEMS and Biomedical Devices
Dr. Monize Caiado Decarli (UMCG) Assistant Professor of Bioprinting & Biomaterials
Dr. Daan Pouwels (UMCG) Assistant Professor of Pulmonary Diseases
In designing these new implants, the team needs fundamental knowledge about materials, but they must also be aware of the fact that surgeons must be able and willing to use them. And they need to be convinced that the new soft materials are strong enough to withstand the pressure inside a blood vessel, or inside the airways of the lungs. ‘The concern over the safety of such innovations is legitimate,’ says Van Rijn. ‘However, decades of experience with soft implants shows that they remain intact inside the body.’
The project also includes even more ambitious innovations: the introduction of smart and active materials. Kamperman: ‘One example would be to add sensors to the implants, or to add drugs to the hydrogel, which are then released over time.’ Other sub-projects look into integrating implants with body tissues, for example by using tissue-compatible glue. Kamperman: ‘These are longer-term goals. First, we have to develop more compatible implants and find out how to place them using minimally invasive techniques.’
Read more about four research projects aimed at developing soft and smart stents and valves below.
Designing new materials with the computer

Assistant Professor Andrea Giuntoli looks at the interface where implant and tissue meet. Since it is very hard to study their interactions through experiments, he uses molecular dynamics simulations. He creates digital models of the interface using the rules of physics and the input of chemists. ‘My speciality is to simulate the mechanics and deformation that take place inside the body on a scale of nanometres,’ he explains. An important part of his work is to study how firmly implants are embedded. ‘We simulate how much force would dislodge it.’
His simulations can take days or even weeks for a computer to process. The simulation results are checked together with chemists and verified through experiments. ‘Because there could, of course, be something wrong with the model.’ He is currently simulating how a lung implant could adhere to the mucus inside the airways. ‘The aim is to design an implant that can glue itself to this mucus layer.’ His work on computer simulations is embedded in both experimental and clinical research. ‘We can discuss implants with a theoretical physicist as well as with the head of a surgery department. That is a really fun part of this job.’
Placing stents using magnets
PhD student Adriana Vasi has to figure out how a stent could be guided through blood vessels to its destination with the use of magnetism. ‘Stent placement is now performed using a plastic catheter. We want to make it less invasive.’ The idea is to inject a small quantity of dehydrated hydrogel, guide it to the right place using magnetism, and induce it to take up water there. ‘This will expand the tubular hydrogel, so that it gets firmly stuck there.’ To achieve this, she uses a gel with thermo-responsive polymers, which is rehydrated by heating it to above body temperature.
Guiding it to its destination is challenging. ‘Magnetic particles are already used in medical studies, but it was tricky to add them to the hydrogel.’ She shows a video clip in which she uses a magnet to guide the stent through a blood vessel model with a weak spot, made from tubing. ‘I had to wiggle the stent with the magnet to get it into position.’ A dehydrated stent, which is smaller, moves more smoothly through the model. In patients, the journey of the stent will be tracked using ultrasound scans. ‘The current placement method with a catheter is tracked with X-rays, which is less safe.’ Vasi has been working on the project for a year now. ‘We need a proof of concept, and then we might move on to using a more realistic model, such as a pig artery.’
Valves for the lung
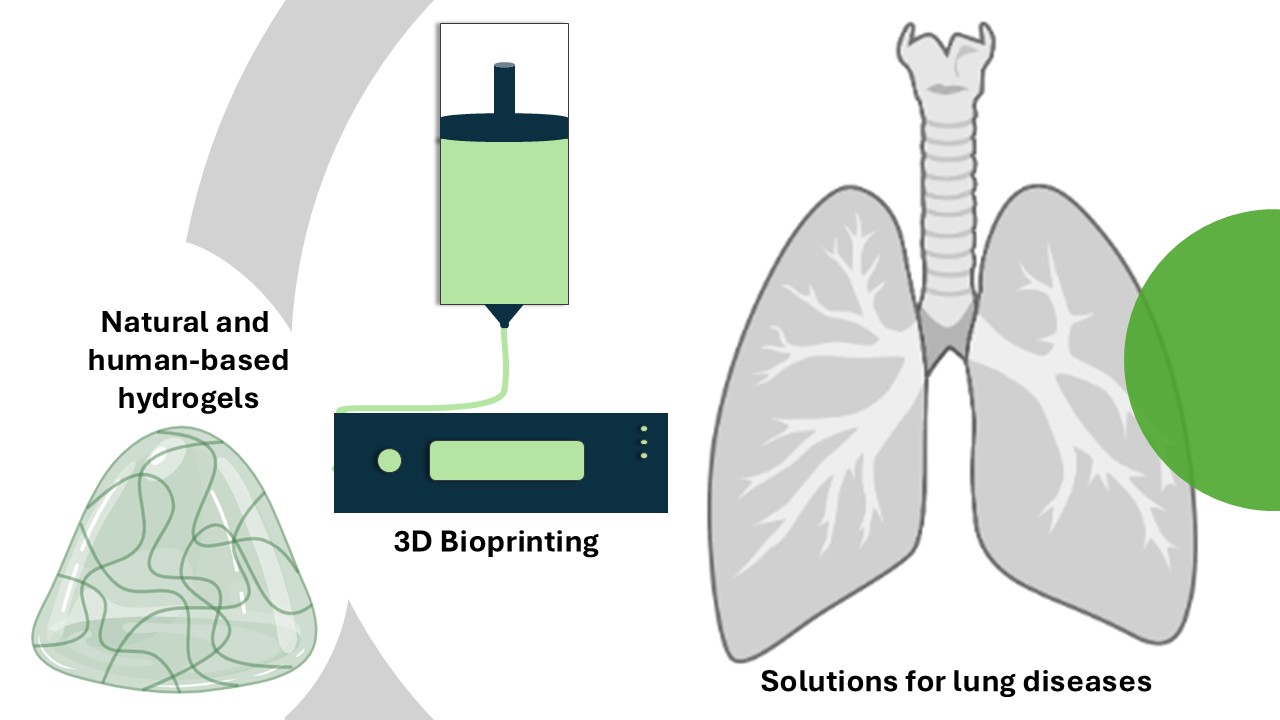
Assistant Professor Monize C. Decarli leads the Bioprinting and Biofabrication Group at the Department of Biomaterials and Biomedical Technology (BBT), UMCG. She recently started to develop 3D printed valves to regulate airflow in the lungs of patients with chronic obstructive pulmonary disease (COPD). Characteristic of this disease is that air gets trapped in parts of the lungs, leading to hyperinflation and a reduction of the functional lung capacity, which means patients have a reduced ability to breathe. By placing a valve in the airways leading to the lung region with trapped air, the air can get out, which alleviates lung hyperinflation.
Decarli: ‘However, the current lung devices have to be replaced in a significant number of cases because of device-induced reactions of the immune system.’ Together with two PhD students and a supervisory team that brings together Dr Małgorzata (Gosia) Włodarczyk-Biegun (UG) and Dr Simon Daan Pouwels (UMCG), Decarli is looking for materials that are more compatible with lung tissue. Her particular focus is on natural hydrogels from biological materials, such as polysaccharides or proteins from human sources. ‘But we are still in the design stage.’ She is thinking outside the box to employ these natural or humanized hydrogels to provide solutions for lung diseases, and not just to mimic the natural valves.
Decarli and her team are 3D printing soft hydrogels to be placed in the airways, which will be combined with other materials to form a final stiffer valve. ‘The final valve will support the airways and should be well tolerated by the body.’ The placement of the valves is another challenge her team will tackle. ‘We are building an experimental setup to test new valves in the lab.’ The team works closely with interventional pulmonologist, Prof. Dr. Dirk-Jan Slebos (UMCG), who will also eventually place the innovative valves.
Modelling flow and friction
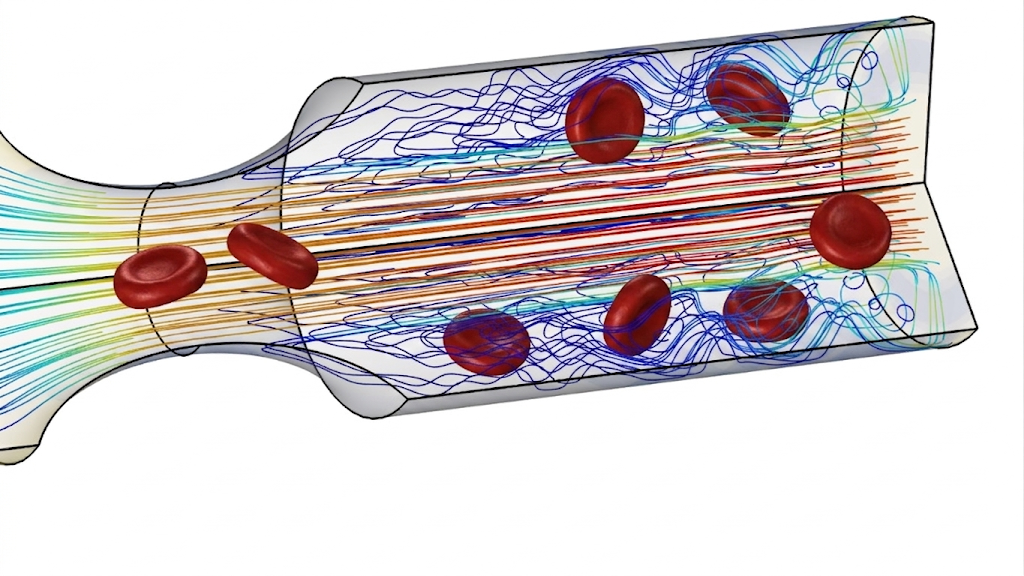
Tim Kousemaker, researcher at the Computational Mechanical and Materials Engineering group (FSE) uses his experience in chemical engineering and computational fluid dynamics to model how stents in blood vessels or bronchi can alter the flow of blood or air. ‘Doctors sometimes see that downstream from a stent that was placed to open a clogged artery, the formation of a new clot appears due to interactions between the stent and the artery wall, as well as plaque deposition.’
Whereas Andrea Giuntoli (see above) models the stents on a molecular level, Kousemaker zooms out and models larger volumes. ‘I use the detailed simulations from Giuntoli as input for my own model.’ Stents also interact with the wall of the blood vessel or airway in which they are placed. ‘There will be some movement on the micrometre level and this causes friction,’ says Kousemaker. ‘My job is to connect different disciplines, such as chemistry, engineering, and immunology. I try to model all these effects with continuum mechanics, a technique that deals with deformable bodies.’
One of his goals is to test new stent designs using his computer model, to see if they are better compared to a benchmark stent. ‘For example, I should be able to calculate if better adhesion can reduce movement and thus diminish friction.’
