Antimicrobial peptides in action
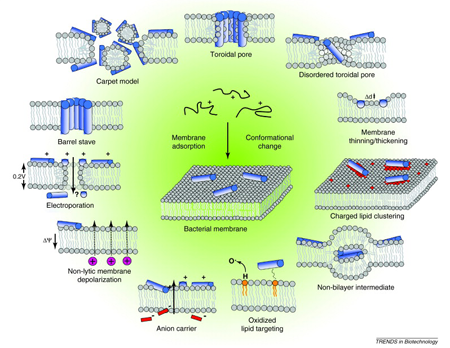
Antimicrobial peptides (AMPs) exhibit a wide range of antimicrobial and antifungal activity and have attracted significant interest as potential antibiotics. However, the mechanism of action of antimicrobial peptides is still poorly understood and, from experimental data, different membrane disruption structural models have been proposed for linear α-helical peptides (the barrel-stave and the toroidal pore models).
We investigate the mechanisms of AMPs in action at different levels of detail: from atomistic [1, 2] to coarse grained molecular dynamics (MD) simulations [3, 4] and even at mixed detail between the two [5]. We proposed a new mechanism for membrane poration by linear antimicrobial peptides: the disordered toroidal pore [1, 2]. Besides, we were able to provide detailed insight on the mechanism of pore-forming cyclic peptides in an effort that brought together simulation results and in vitro measurements of peptide activity [6].
[1] H. Leontiadou, A.E. Mark, S.J. Marrink. Antimicrobial peptides in action. J. Am. Chem. Soc. 128:12156–12161, 2006.
[2] D. Sengupta, , H. Leontiadou, A.E. Mark, S.J. Marrink. Toroidal pores formed by antimicrobial peptides show significant disorder. Biochim. Biophys. Acta. 1778:2308–2317, 2008.
[3] A. J. Rzepiela, D. Sengupta, N. Goga, S.J. Marrink. Membrane poration by antimicrobial peptides combining atomistic and coarse-grained descriptions. Faraday Discuss., 144:431–443, 2010.
[4] A.A. Polyansky, R. Ramaswamy, P.E. Volynsky, I.F. Sbalzarini, S.J. Marrink, R.G. Efremov. Antimicrobial peptides induce growth of phosphatidylglycerol domains in a model bacterial membrane. J. Phys. Chem. Lett., 1 (20): 3108–3111, 2010
[5] T.A. Wassenaar, H. I Ingólfsson, M. Prieß, S.J. Marrink, L.V Schäfer, Mixing MARTINI: Electrostatic coupling in hybrid atomistic-coarse-grained biomolecular simulations. J. Phys. Chem. B, 117 (13):3516–3530, 2013.
[6] A.D. Cirac, G. Moiset, J.T. Mika, A. Koçer, P. Salvador, B. Poolman, S. J. Marrink, D. Sengupta. The molecular basis for antimicrobial activity of pore-forming cyclic peptides.Biophys. J. 100(10):2422-31, 2011.
| Last modified: | 02 July 2015 2.14 p.m. |
