
1. Mechanisms involved in HSC self-renewal and hematopoietic lineage commitment
Hematopoiesis is initiated by a relatively small number of multipotent self-renewing hematopoietic stem cells that generate large numbers of differentiated progeny by a process of amplification and progressive lineage restriction. The process of hematopoietic differentiation involves the generation of multilineage progenitors that are restricted to either the myeloid or lymphoid lineages (the CMP, common myeloid progenitor and the CLP, common lymphoid progenitor, and further differentiation of these pluripotent cells generates mature erythroid, megakaryocytic, myeloid or lymphoid cells (Fig.1).
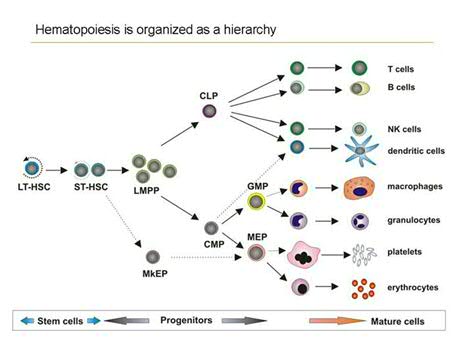
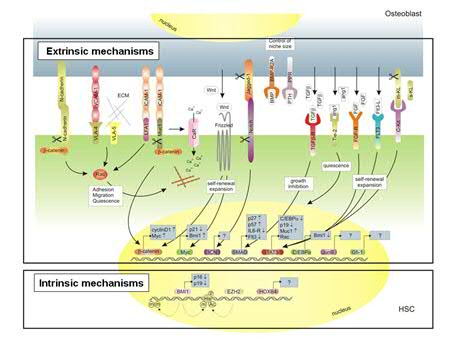
HSC self-renewal and hematopoietic differentiation are tightly controlled by multiple positive and negative regulatory elements, but mechanisms modulating the balance between self-renewal and entrance into a differentiation program are poorly understood. Both intrinsic as well as extrinsic mechanisms are likely to be involved in the regulation of hematopoietic stem cell self-renewal. Intrinsic mechanisms include those that regulate the epigenetic status of HSCs, eg mediated via the Polycomb Repression Complexes (PRC) 1 and 2, while extrinsic pathways include cues that are dictated by the stem cell microenvironment (Fig.2). The relative contribution of these intrinsic and extrinsic pathways to human hematopoietic stem cell self-renewal is studied in detail in our lab.
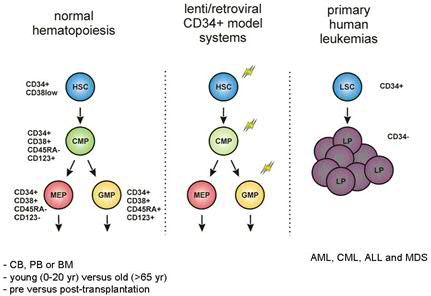
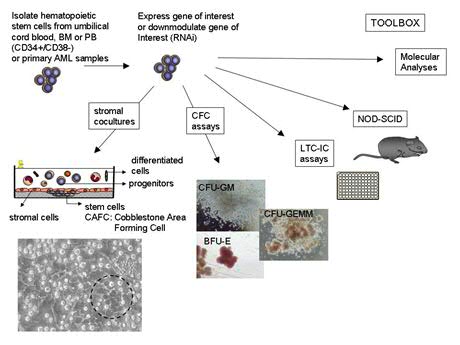
In order to be able to study these pathways in detail, we make use of retrovriral and lentiviral transduction procedures to stably express or downregulate genes. The various retroviral model systems that we have summarized in figure 3 and an overview of our toolbox is shown in figure 4. Primary human hematopoietic stem and progenitor cells that we study are isolated from CB, PB or BM. We are interested in differences between fetal, yound and old HSCs and MPPs, and in differences between HSCs and MPPs pre and post-transplantation in patients. Furthermore, we are making use of primary stem and progenitor cells isolated from PB or BM of leukemia patients (Fig.5)

Even though the NSG xenograft mouse model is often considered to be the gold standard for human hematoppoiesis/leukemia studies, these mice are rather B-cell lymphoid-biased and many patient samples either do not engraft at all or fail te repopulate secondary reciepients due to loss of stemness. Most likely this is related to the murine bone marrow niche, which does not provide the essential human growth factors, cytokines and adhesion factors that are required for efficient maintenance of stemness. Therefore, we have begun to to generate human niche in xenograft mouse models in order to better recapitulate human stem cell biology and leukemogenesis in vivo. By making use of primary Mesenchymal Stem Cells (MSCs) that we implant subcutaenously in NSG mice we have been able to recreate a human niche in the mouse. For further info, please see:
Establishing human leukemia xenograft mouse models by implanting human bone marrow-like scaffold-based niches. Antonelli A, Noort WA, Jaques J, de Boer B, de Jong-Korlaar R, Brouwers-Vos AZ, Lubbers-Aalders L, van Velzen JF, Bloem AC, Yuan H, de Bruijn JD, Ossenkoppele GJ, Martens AC, Vellenga E, Groen RW, Schuringa JJ. Blood. 2016 Dec 22;128(25):2949-2959. doi: 10.1182/blood-2016-05-719021. Epub 2016 Oct 12.
Genetically engineered mesenchymal stromal cells produce IL-3 and TPO to further improve human scaffold-based xenograft models. Carretta M, de Boer B, Jaques J, Antonelli A, Horton SJ, Yuan H, de Bruijn JD, Groen RWJ, Vellenga E, Schuringa JJ. Exp Hematol. 2017 Jul;51:36-46. doi:0.1016/j.exphem.2017.04.008. Epub 2017 Apr 26.
Modeling BCR-ABL and MLL-AF9 leukemia in a human bone marrow-like scaffold-based xenograft model. Sontakke P, Carretta M, Jaques J, Brouwers-Vos AZ, Lubbers-Aalders L, Yuan H, de Bruijn JD, Martens AC, Vellenga E, Groen RW, Schuringa JJ. Leukemia. 2016 Oct;30(10):2064-2073. doi: 10.1038/leu.2016.108. Epub 2016 Apr 29.
For more information on these projects, please contact J.J.Schuringa: j.j.schuringa@umcg.nl
