News
October 2021
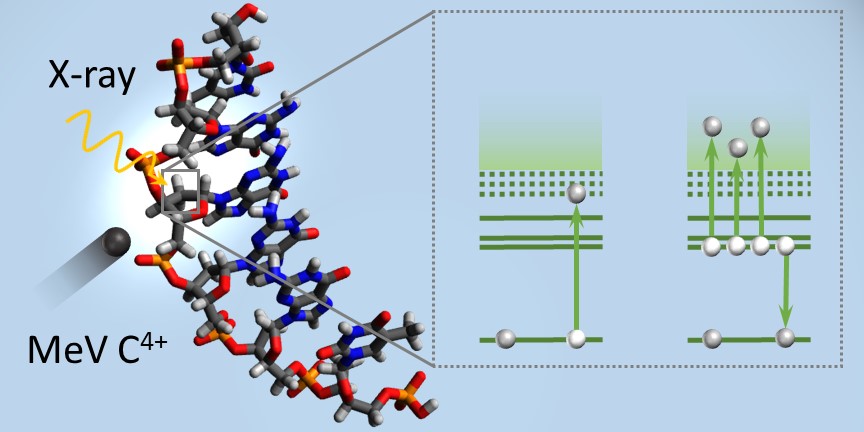
In her PhD project supported by the China Scholarship Council (CSC), Wen Li works on the molecular mechanisms underlying biological radiation damage. Wen has experimentally demonstrated that the inner-shell vacancies induced by X-ray and heavy ion irradiation of DNA are filled by Auger decay under emission of several secondary electrons which has profound implications for modeling of radiation damage. Wen did her experiments at the BESSY II synchroton in Berlin, Germany and at the GANIL IRRSUD facility in Caen, France. The experimental setup was developed at the Zernike Institute and interfaced with the X-ray and heavy-ion beamlines. Complementary experiments with femtosecond infrared laser-pulses have been performed in Markus Gühr’s laboratory in Potsdam. Wen’s work has just been published as an Edge Article in Chemical Science.
June 2021
Xin Wang works on his PhD project (supported by the China Scholarship Council, CSC) focusing on the interaction of soft X-rays with gas-phase DNA. Experiments are performed at the BESSY II synchroton in Berlin, either with our own apparatus developed at the Zernike Institute, or with the Ion Trap setup of Tobias Lau's group. The interpretation of the experimental data is only possible with the help of quantum chemical calculations which are performed in close collaboration with Shirin Faraji and her group. In this publication, Xin shows for the first time how the site-selectivity of soft X-ray absorption can be employed to study the protonation sites in gas-phase oligonucleotides. This work has just been selected by Physical Chemistry - Chemical Physics as one of their HOT ARTICLES in 2021.

February/March 2020
In an international collaboration with colleagues from Potsdam/Germany, Hamburg/Germany, Caen/France and Gothenburg/Sweden, we have investigated structural dynamics in self assembled DNA nanostructures at the free electron laser FLASH 2 in Hamburg. We will not forget this beamtime for a long time, as we were the last users before the COVID19-related shutdown and our last shift ended on March 15.
April 1 2019

September 12 2018: Atomic hydrogen interactions with gas-phase coronene cations: hydrogenation versus fragmentation

In a fruitful collaboration between theory (Mathias Rapacioli and Aude Simon from University of Toulouse) , astronomy (Stephanie Cazaux, Delft Universty of Technology) and experiments (our team here at the Zernike Institute), we have studied the response of trapped gas-phase coronene cations upon interaction with hydrogen atoms. The results have just been published in PCCP and are hightlighted on the journal cover, which shows an artist impression of the H-induced dissociation of a coronene cation.
Abstract:
Sequential hydrogenation of polycyclic aromatic hydrocarbon (PAH) cations drives a gradual transition from a planar to a puckered geometry and from an aromatic to an aliphatic electronic structure. The resulting H-induced weakening of the molecular structure together with the exothermic nature of the consecutive H-attachment processes can lead to substantial molecular fragmentation. We have studied H attachment to gas-phase coronene cations in a radiofrequency ion trap using tandem mass spectrometry. With increasing hydrogenation, C2Hi loss and multifragmentation are identified as main de-excitation channels. To understand the dependence of both channels on H-exposure time, we have simulated the molecular stability and fragmentation channels of hydrogenated PAHs using a molecular dynamics approach employing potential energies determined by a density functional based tight binding method. As the coronene fragmentation patterns depend on the balance between energy deposition by H-attachment and the extent of cooling in between subsequent attachment processes, we investigate several scenarios for the energy distribution of hydrogenated PAHs. Good agreement between experiment and simulation is reached, when realistic energy distributions are considered.