Research Julia Kamenz
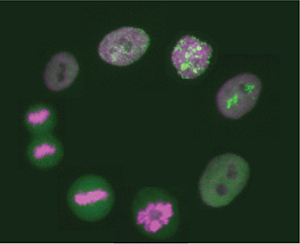
Every day billions of cells divide in our bodies. Errors during cell division can lead to the loss or gain of genomic information and result in cell death or disease development, such as cancer. We are using a combination of biochemistry, quantitative cell biology and computational modeling to unravel the complex molecular mechanisms underlying cell division and ensuring error-free cell proliferation.
Currently, we are focusing on two main topics:
(1) How phosphorylation events help to establish temporal order during cell cycle progression.
(2) How phosphatases contribute to the regulation of cell cycle progression.
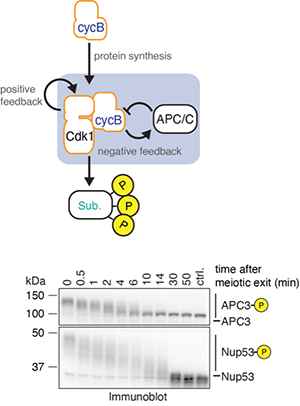
The temporal dynamics of the cell cycle
The early embryonic divisions in many organisms, including the African clawed frog Xenopus laevis, are very fast and highly robust - almost clock-like. Despite our detailed knowledge about most of the key players involved in this process, we still do not understand how these rapid cell divisions and all accompanying processes can be executed with such temporal precision.
Specifically, we are interested in how the hundreds -if not thousands - of phosphorylation events mediated by the cyclin B-Cdk1 complex during mitosis translate into the distinct order of mitotic events necessary for error-free cell division.
To answer this question, we combine biochemical assays using frog egg extracts, which amazingly retain the capacity to undergo the biochemical changes associated with cell cycle progression ex vivo, with quantitative mass spectrometry. This approach will reveal new insights into how, when and in which order proteins become phosphorylated during mitosis and how these phosphorylations regulate protein function.
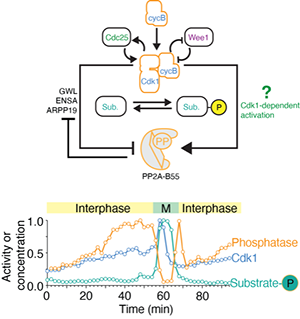
Cell cycle regulation by phosphatases
For a long time, phosphatases have been considered to be the unspecific and unregulated counterparts to the sophisticated regulation of cell cycle kinases. However, this picture has dramatically changed, demonstrating that phosphatases are highly specific and highly regulated throughout the cell cycle. We are particularly interested in how signaling pathways directly modulate phosphatase activity in order to regulate cell cycle progression. Currently, we are focusing on the phosphatase PP2A-B55, which we recently showed is both inhibited by Cdk1 through double-negative feedback but also activated by an unknown Cdk1-dependent mechanism constituting an incoherent feedforward loop (Kamenz et al., 2021).
Using both, biochemical approaches employing frog egg extracts as well as live cell imaging of human cells in cell culture, we will study how Cdk1 activity and cell cycle signaling in general impinge on the activity of PP2A-B55 and other phosphatases.

Further reading
Julia Kamenz, Lendert Gelens and James E Ferrell Jr. Bistable, biphasic regulation of PP2A-B55 accounts for the dynamics of mitotic substrate phosphorylation Current Biology, 2021 Feb 22; 31(4):794-808
Sarah Lockhead, Alisa Moskaleva, Julia Kamenz, Yuxin Chen, Minjung Kang, Anay R Reddy A, Silvia DM Santos, James E Ferrell Jr. The Apparent Requirement for Protein Synthesis during G2 Phase Is due to Checkpoint Activation Cell Reports, 2020 July 14; 32(2):107901
Julia Kamenz and James E Ferrell Jr.* The temporal ordering of cell-cycle phosphorylation Molecular Cell, 2017 Feb 2; 65(3): 371-373
Julia Kamenz, Tamara Mihaljev, Armin Kubis, Stefan Legewie and Silke Hauf* Robust ordering of anaphase events by adaptive thresholds and competing degradation pathways
