Elektronen opzuigen met zuur
Aromatische verbindingen vind je veel in de natuur, maar ook chemische fabrieken maken er jaarlijks miljoenen tonnen. ‘Aromatisch’ betekent hier niet dat ze lekker ruiken, maar het verwijst naar de aanwezigheid van een ringstructuur van koolstofatomen in het molecuul. Dit soort structuren is erg stabiel en helemaal plat. Er bestaan ook niet-aromatische ringstructuren die niet plat zijn. Adjunct-hoogleraar organische chemie Syuzanna Harutyunyan ontving deze week een ERC Consolidator Grant van 2 miljoen euro om een methode te ontwikkelen waarmee 3D structuren zijn te maken van 2D ringen.

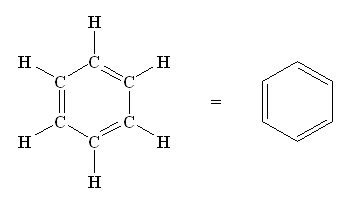
Het klassieke voorbeeld van een aromatische verbinding is benzeen. Dit is een zeshoekig molecuul gemaakt van zes koolstofatomen waar steeds een waterstofatoom aan vast zit. De ring is stabiel vanwege de vrije elektronen die aan de binnenkant zitten – het kenmerk van een aromatische verbinding. Er zijn allerlei chemische reacties bekend met dit soort aromatische ringen waarbij ze intact blijven, maar vanwege de grote stabiliteit zijn reacties die de ring fundamenteel veranderen veel lastiger. ‘Maar driedimensionale moleculen die bestaan uit een aantal verbonden niet-aromatisch ringen zijn belangrijk voor het namaken van natuurlijke producten en geneesmiddelen’, legt Harutyunyan uit.
‘Organisch chemici moeten dit soort verbindingen uit losse koolstofatomen opbouwen. Dat is jammer, het zou veel eenvoudiger zijn om aromatische ringen als bouwsteen te gebruiken, maar je kunt ze niet zomaar veranderen in niet-aromatische ringen.’ Zowel de aromatische als de niet-aromatische ringen zijn zeer stabiel, maar om de een in de ander te veranderen moet je de ring eerst destabiliseren en daarna verbouwen. Dit proces het ook wel de-aromatiseren en het zou een efficiënte manier kunnen zijn om de eenvoudige platte aromatische verbindingen te veranderen in de complexe structuren waaruit geneesmiddelen bestaan.
‘Dit is precies wat ik in mijn project wil doen’, vertelt Harutyunyan zelfverzekerd. Ze heeft al wat experimenten gedaan waaruit blijkt dat haar idee echt werkt. Om de aromatische ring te destabiliseren gebruikt zij een zogeheten Lewiszuur. Deze Lewiszuren zijn chemische stoffen die vrije elektronenparen aantrekken. Door elektronen ‘weg te trekken’ kan een Lewiszuur reactiever maken. Dit concept is al in 1923 beschreven door de Amerikaanse fysisch-chemicus Gilbert N. Lewis.
‘Maar iedereen dacht dat de zuren niet sterk genoeg zijn om een aromatische ring te destabiliseren’, legt Harutyunyan uit. Maar zij heeft onlangs al een Lewiszuur gebruikt om een andere ‘onmogelijke’ reactie uit te voeren en zij dacht dat ze dit trucje ook wel zouden kunnen doen. ‘Ik vond een artikel uit 1985, dat een structuur beschreef gemaakt van een Lewiszuur en een aromatische ring. Dat overtuigde mij ervan dat het zuur met de ring kon reageren.’
Het Lewiszuur zuigt de stabiliserende elektronen uit de ring. Dit maakt het eenvoudiger de aromatische structuur te reduceren tot een niet-aromatische ring. ‘Een promovendus uit mijn groep heeft een paar experimenten gedaan en het werkt. Nu is het zaak om het precies zo te laten werken als ik wil.’ De komende vijf jaar zullen drie promovendi en wat postdocs uitzoeken hoe specifieke moleculen zijn te maken van aromatische bouwstenen.
‘Uiteindelijk zou dit werk ook voor een beter gebruik van biomassa kunnen zorgen’, zegt Harutyunyan. Lignine, een taaie vezel die planten hun stevigheid geeft, is bijvoorbeeld een polymeer van aromatische moleculen. Wanneer je lignine afbreekt tot kleinere aromatische verbindingen zou je deze met de processen die het ERC project zal ontwikkelen kunnen gebruiken om allerlei nuttige chemische verbindingen te maken.
Meer nieuws
-
14 januari 2026
Wat de geur van de zee doet met de wolken boven Antarctica
-
13 januari 2026
Atomair model verklaart hoe geheugenmetalen van vorm veranderen
