Sensor meet drukte in een cel
Wetenschappers van de Rijksuniversiteit Groningen hebben een moleculaire sensor ontwikkeld waarmee ze ‘drukte’ in een cel kunnen meten die veroorzaakt wordt door de aanwezigheid van grote moleculen. Met de sensor is het mogelijk de concentratie van dit soort moleculen in zowel bacteriën als in zoogdiercellen kwantitatief te meten. Een beschrijving van de werking van de sensor is op 2 februari 2015 gepubliceerd door het tijdschrift Nature Methods.
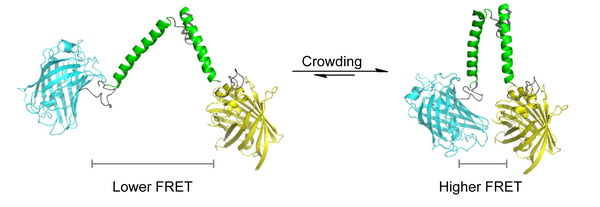
Levende cellen zitten tjokvol grote moleculen zoals eiwitten en nucleïnezuren. Dit beïnvloedt de manier waarop grote en kleine moleculen in een cel met elkaar reageren. De drukte (crowding genoemd) vermindert de snelheid waarmee deeltjes diffunderen, maar zorgt er ook voor dat moleculen vlak bij elkaar blijven zitten.
Eindelijk meetbaar
De transcriptie van DNA verloopt onder realistische condities zoals die in de cel aanwezig zijn vele malen sneller dan in meer verdunde condities die tijdens laboratoriumexperimenten gebruikelijk zijn. Toch houden onderzoekers bij het beschrijven en ‘engineeren’ van processen in de cel zelden rekening met crowding. Een belangrijke reden daarvoor is dat er geen goede techniek is om te meten hoe groot de drukte in cellen precies is.
Tot nu toe maken onderzoekers schattingen, gebaseerd op de gemiddelde concentratie van macromoleculen en het gemiddelde volume van de cellen. Met de sensor die nu ontwikkeld is, kan eindelijk direct gemeten worden hoe sterk de crowding in cellen precies is. Het is zelfs mogelijk zichtbaar te maken hoe de drukte zich ontwikkelt in de tijd in verschillende delen van de cel.
‘Veer’ van eiwit
De sensor is ontwikkeld door dr. Arnold Boersma en hoogleraar biochemie Bert Poolman. Zij ontwikkelden een ‘veer’ van eiwit met fluorescente eiwitgroepen die beide einden markeren. De eerste eiwitgroep zendt blauw licht uit wanneer het door een laser wordt beschenen. Het blauwe licht slaat de tweede eiwitgroep aan dat vervolgens geel licht gaat uitzenden. De overdracht van energie is recht evenredig met de afstand tussen beide groepen. Deze techniek heet ‘Förster resonance energy transfer’ (FRET).
Mechanische druk
Macromoleculen oefenen een mechanische druk uit op de veer, die beide fluorescente groepen op de uiteinden dichter bij elkaar brengt. Een serie controle experimenten laat zien dat andere krachten (zoals de ionsterkte of chemische affiniteit) geen invloed hebben op de afstand tussen beide fluorescente groepen. Andere experimenten lieten zien dat de sensor een nauwkeurige kwantitatieve meting van de drukte in een cel oplevert.
Kunstmatig gen
Er is een kunstmatig gen gemaakt dat zorgt voor de aanmaak van de sensor in cellen. Er zijn twee versies van: één voor bacteriële cellen en één voor zoogdiercellen. ‘Wij zullen de sensor gebruiken om de structuur van het cytoplasma tijdens de celcyclus in kaart te brengen’, zegt Poolman. ‘Onze interesse gaat uit naar de manier waarop cellen hun volume kunnen regelen, iets dat natuurlijk de mate van crowding in de cel beïnvloedt. Maar er zijn nog tal van andere mogelijke toepassingen voor deze sensor.’
Andere toepassingen
Informatie over de drukte in cellen is bijvoorbeeld van belang voor vakgebieden als Systeem Biologie en Synthetische Biologie, omdat de omstandigheden in een cel invloed hebben op de snelheid van biochemische reacties en de affiniteit tussen biologische moleculen, maar ook op de vouwing van eiwitten in levende cellen. ‘We willen weten hoe cellen werken en hoe we ze kunnen aanpassen en ontwerpen om ze een specifieke functie te geven’, zegt Poolman, die tevens programmadirecteur is van het Centrum voor Synthetische Biologie aan de RUG. Hij verwacht dat de nieuwe sensor zal bijdragen aan een beter begrip van de manier waarop cellen functioneren.
Meer informatie
-
Contact: prof.dr. B. (Bert) Poolman
-
De sensor is aan de RUG ontwikkeld binnen het Groningen Biomolecular Sciences and Biotechnology Institute en het Zernike Institute for Advanced Materials.
-
Referentie: Boersma, A.J.; Zuhorn, I.S.; and Poolman, B. ; A sensor for quantification of macromolecular crowding in living cells in Nature Methods DOI 10.1038/nmeth.3257
Meer nieuws
-
09 juni 2026
Rastogi ontvangt Heineken Young Scientists Award
-
08 juni 2026
Duurzaam samenleven op de savanne
