Nature Chemistry: Groningse chemici maken schakelbaar antibioticum
Wetenschappers van de Rijksuniversiteit Groningen hebben een antibioticum gemaakt waarvan de activiteit is te controleren met licht. Het is mogelijk dit middel vlak voor gebruik ‘aan’ te zetten, waarna het langzaam zijn activiteit verliest. Op die manier is te voorkomen dat er werkzaam antibioticum in het milieu terechtkomt. Deze vondst is zondag 15 september 2013 gepubliceerd op de website van het wetenschappelijke tijdschrift Nature Chemistry.
Antibiotica beschermen ons tegen dodelijke infecties. Ook in de agrarische sector worden antibiotica op ruime schaal gebruikt. Maar na gebruik komen ze, bijvoorbeeld via de riolering, in het milieu. Daar kunnen ze het ontstaan van resistente bacteriestammen bevorderen.
Met die gedachte gingen de Groningse onderzoekers kijken of er een antibioticum valt te maken waarvan de activiteit is te controleren. Wanneer er alleen geïnactiveerde antibiotica in het milieu terechtkomen is er immers geen gevaar voor resistentie.
Varianten
Hoogleraar organische chemie Ben Feringa is pionier op het terrein van door licht geactiveerde moleculaire schakelaren. Zijn promovendus Willem Velema maakte varianten van een antibioticum waar een met licht schakelbare groep aan werd gezet. ‘'Het is lastig te voorspellen wat er gebeurt met de activiteit als een groep in het molecuul veranderd wordt’, legt Velema uit. De nieuwe groep beïnvloedt immers de werking van het antibioticum. ‘En dat moet precies op de juiste manier gebeuren.’
Velema maakte negen varianten, waarvan er één zeer goed bleek te werken in testen die samen met de groep van moleculair bioloog Arnold Driessen werden uitgevoerd. De schakelaar zorgt er voor dat het antibioticum in de normale stand niet werkt. Maar na bestraling met ultraviolet licht klapt een deel van het schakelmolecuul om, waardoor het antibioticum weer actief wordt. In chemische termen: de schakelgroep gaat over van een trans- naar een cis-isomeer.
Halfwaardetijd
In het Nature Chemistry artikel laten de onderzoekers zien dat het mogelijk is het antibioticum op elk gewenst moment aan te zetten. Vervolgens valt het molecuul vanzelf terug van de actieve cis-isomeer naar de niet actieve trans-isomeer. Bij lichaamstemperatuur is de halfwaardetijd ongeveer twee uur.
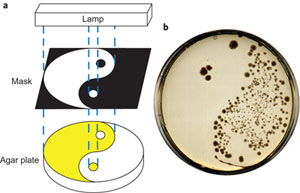
Ben Feringa benadrukt dat de experimenten alleen bedoeld zijn om te laten zien dat het mogelijk is de werkzaamheid van een antibioticum te controleren met licht. ‘De weg van dit idee naar een werkend geneesmiddel is erg lang, dat kan nog wel tien jaar duren.’ Behalve dat een schakelbaar antibioticum het milieu niet belast, zou je het ook kunnen gebruiken om heel gericht plekken in het lichaam te behandelen. Feringa: ‘Voor een infectie in de huid kan je een onwerkzaam antibioticum slikken en dat op de plaats waar het nodig is activeren. Dan dood je geen nuttige darmbacteriën.’
Bacteriegroei
Voorlopig is het schakelbare antibioticum vooral handig voor onderzoek. ‘Het is mogelijk de groei van bacteriën heel gericht op één plek en op een specifiek moment te remmen. ’ Je zou zo bijvoorbeeld kunnen onderzoeken welke factoren een invloed hebben op de groei van de bacterie en de werkzaamheid van het antibioticum of het ontstaan van resistentie. ‘Maar onze belangrijkste boodschap met dit artikel is dat het mogelijk is om de werking van een geneesmiddel, dat bestaat uit een relatief klein molecuul, extern te controleren, in dit geval met licht. Dat is een hele nieuwe manier om over geneesmiddelenonderzoek na te denken’, aldus Feringa.
Meer informatie:
- prof. dr. Ben Feringa
- drs. Willem Velema
Optical control of antibacterial activity. Willem A. Velema1, Jan Pieter van der Berg2, Mickel J. Hansen1, Wiktor Szymanski1, Arnold J. M. Driessen2,3 and Ben L. Feringa1,3. Nature Chemistry, online 15 september (DOI: 10.1038/nchem.1750)
- Centre for Systems Chemistry, Stratingh Institute for Chemistry, University of Groningen, Nijenborgh 4, 9747 AG, Groningen, The Netherlands
- Molecular Microbiology, Groningen Biomolecular Sciences and Biotechnology Institute, University of Groningen, Nijenborgh 7, 9747 AG, Groningen, The Netherlands
- Zernike Institute for Advanced Materials, University of Groningen, Nijenborgh 4, 9747 AG, Groningen, The Netherlands
Dit onderzoek is mogelijk gemaakt door financiële ondersteuning van NWO (Nederlandse Organisatie voor Wetenschappelijk Onderzoek), KNAW (Koninklijke Nederlandse Akademie voor Wetenschappen), ERC (European Research Council) en het Ministerie van OC&W (Zwaartekracht programma).
