RUG-chemici maken ‘onnatuurlijk’ enzym met nieuwe mogelijkheden
Boorzuur komt in geen enkel organisme voor, maar is toch al tientallen jaren in gebruik in de organische chemie. ‘Daarmee zijn namelijk chemische reacties uit te voeren die we niet in de natuur zien’, legt Gerard Roelfes uit. Hij is hoogleraar Biomoleculaire Chemie en Katalyse aan de RUG. Zijn onderzoeksgroep maakte een enzym dat boor bevat als reactieve groep. Daarmee is het mogelijk de reacties meer selectief te maken en de katalytische eigenschappen te verbeteren via geleide evolutie. Bovendien zijn enzymatische reacties duurzamer dan gewone chemische reacties, omdat ze bij lage temperatuur en zonder giftige oplosmiddelen plaatsvinden. Het niet-natuurlijke enzym met boorzuur is beschreven in een publicatie die op 8 mei online verscheen bij het wetenschappelijke tijdschrift Nature.
FSE Science Newsroom | René Fransen
Het gebruik van boor bij reacties in de organische chemie begon zo’n zeventig jaar geleden, en in 1979 werd daar een Nobelprijs voor toegekend. De laatste jaren is er een toenemende belangstelling voor boor als katalysator van reacties, maar het gebruik ervan in de chemische industrie is nog beperkt. Roelfes legt uit waarom dat zo is: ‘De reacties die boor katalyseert verlopen nog te traag. En daarnaast is het niet zo geschikt om zogeheten enantioselectieve reacties uit te voeren.’ Dat soort reacties wordt gebuikt om chirale moleculen te maken, verbindingen die bestaan in twee versies die elkaars spiegelbeeld zijn, zoals een linker- en rechterhand. Vooral bij geneesmiddelen kunnen deze twee ‘handen’ een verschillende werking hebben. Daarom is het van belang voor de farmaceutische industrie om heel selectief de juiste ‘hand’ te maken.
(Artikel gaat verder onder de foto)
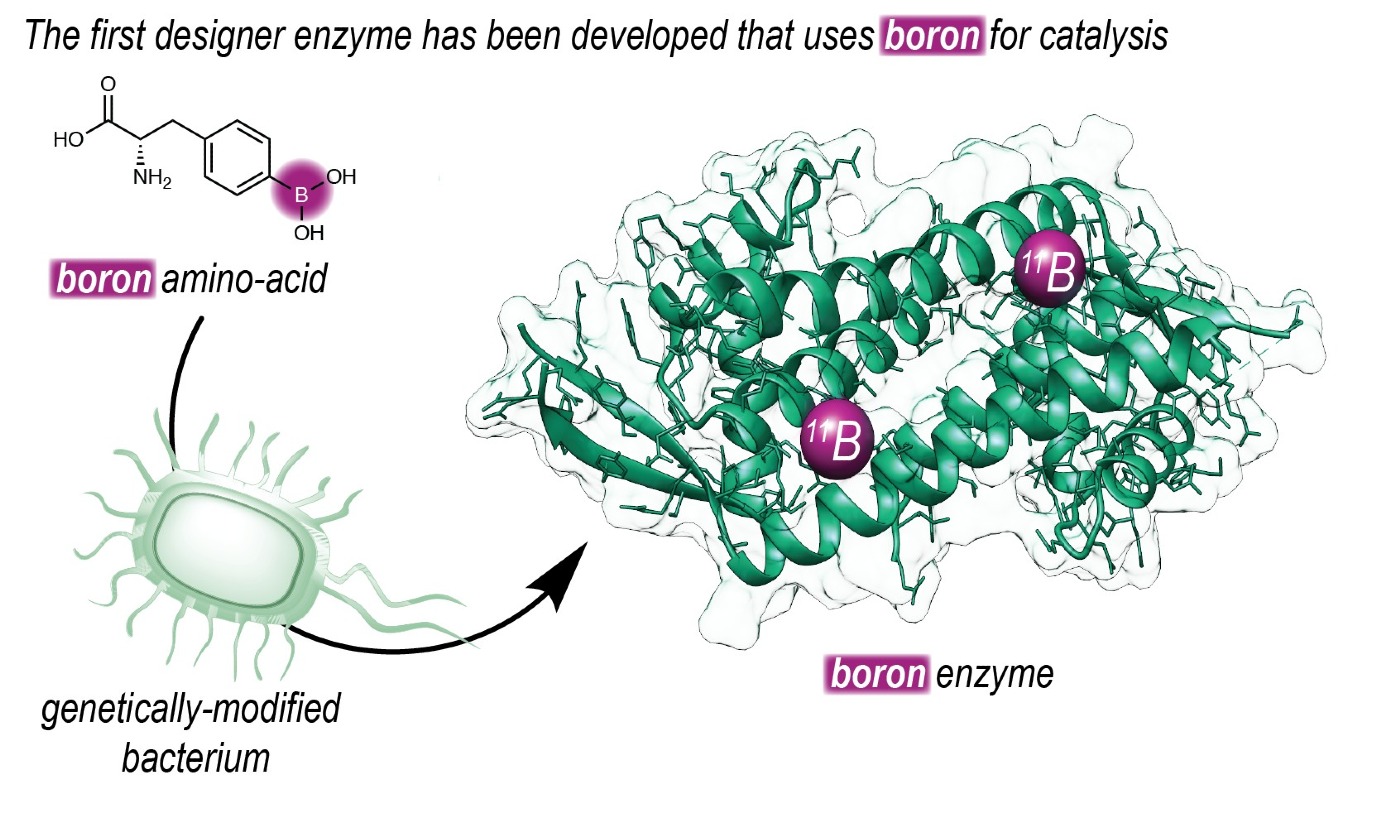
Selectieve reactie
‘Om dit mogelijk te maken hebben we boor in een enzym geplaatst. Onze groep heeft een lange geschiedenis in het ontwerpen van enzymen die niet bestaan in de natuur.’ De groep van Roelfes gebruikt een zogeheten ‘uitgebreide genetische code’ om een aminozuur dat reactief boorzuur boorzuur bevat in een enzym te zetten. ‘Met deze techniek kunnen we heel precies, op DNA niveau, bepalen waar we dit aminozuur in het enzym-eiwit plaatsen.’
Nadat ze een enzym met boorzuur in het reactieve centrum hadden gemaakt konden ze met behulp van geleide evolutie de efficiëntie ervan verhogen, waardoor de reactie sneller wordt uitgevoerd. ‘Bovendien was de reactie heel selectief voor één van de twee ‘handen’, omdat deze nu binnen de chirale structuur van het enzym plaatsvond.’ De reactie, die is beschreven in het Nature-artikel, heeft geen direct nut voor de industrie maar is gebruikt om te laten zien dat het mogelijk is de katalytische eigenschappen van boor te verwerken in een enzym.

Biokatalyse
Het gebruik van enzymen is belangrijk voor onder mee de farmaceutische industrie. ‘In hun zoektocht naar manieren om geneesmiddelen op een groenere een duurzame manier te produceren willen ze graag klassieke chemische reacties vervangen door biokatalyse met enzymen.’ Bij de RUG werken verschillende onderzoekgroepen samen om dit te bereiken. ‘We hebben een aantal onderzoeksgroepen in de Faculty of Science and Engineering die dit soort werk doen, en verschillende manieren gebruiken om biokatalyse in te zetten in de chemische industrie.’ Roelfes en zijn team zullen dan ook verdergaan in het ontwikkelen van enzymen met boorzuur en andere ‘onnatuurlijke’ enzymen.
Referentie: Lars Longwitz, Reuben B. Leveson-Gower, Henriëtte J. Rozeboom, Andy-Mark W. H. Thunnissen & Gerard Roelfes: Boron catalysis in a designer enzyme. Nature, online 8 May 2024.
Meer nieuws
-
26 mei 2026
Babs Gons nieuwe gastschrijver RUG
-
26 mei 2026
Studententeams strijden met zelfrijdende auto’s
