Hoe staat het met de motoren van Feringa?

In december 2016 ontving hoogleraar organische chemie Ben Feringa de Nobelprijs voor zijn werk aan moleculaire motoren. Dit zijn piepkleine machines, niet meer dan een paar miljardste meter groot. Onze cellen zitten vol met complexe moleculaire motoren en Feringa gebruikte die als inspiratie om synthetische varianten te maken, die in één richting konden ronddraaien. Na de toekenning van de Nobelprijs trok Feringa de hele wereld rond om gastcolleges te geven en zijn visie op wetenschap uit te dragen. Maar wat voor interessant onderzoek kwam er sinds het najaar van 2016 uit zijn eigen lab?
De ontdekking van een moleculaire motor kwam min of meer bij toeval. In de jaren 1990 werkte Feringa onder meer met moleculen die konden schakelen tussen twee vormen met behulp van licht. Daarbij ontdekte hij dat een van die synthetische moleculaire schakelaars feitelijk in één richting draaide wanneer die werd blootgesteld aan licht en warmte: het was een roterende motor. Aangezien de motoren en schakelaars met licht zijn te controleren zouden ze bruikbaar zijn in dynamische moleculaire systemen.

Nano-auto
De eerste door licht aangedreven rotatiemotoren vormden de basis voor het werk dat Feringa drie jaar geleden de Nobelprijs voor de Chemie opleverde. Hierop voortbouwend creëerde Feringa allerlei motoren en schakelaars die reageren op licht van verschillende kleuren. Zijn meest iconische bouwsel is de nano-auto. Die auto, slechts een paar nanometer lang, reed over een oppervlak van koper en werd aangedreven door elektronen die uit de punt van een scannning tunneling microscoop kwamen. Maar deze auto rijdt alleen in hoog-vacuüm bij 4 graden Kelvin (dat is -269 graden Celsius!) en heeft een speciaal gebouwd ‘weg’ nodig. Hoewel dit een prestatie van formaat was, zal het nog enorm veel onderzoek kosten om dit soort auto’s klaar te maken voor praktische toepassingen. Eind 2019 promoveerde er weer een onderzoeker bij Feringa op het bouwen van een weg voor nano-auto’s. Daarmee vergeleken liggen toepassingen van moleculaire motoren en schakelaars veel dichterbij. Met behulp van chemische aanpassingen zijn er al verschillende wetenschappelijke toepassingen mee gemaakt. En de afgelopen drie jaar zijn schakelaars ook al bestudeerd en gebruikt buiten de synthetische organische chemie. Het is onmogelijk om alle studies die vanuit het Feringa-lab zijn gepubliceerd te bespreken, daarom behandelt dit artikel alleen in grote lijnen de voortgang in het onderzoek naar moleculaire motoren en schakelaars vanaf 2016.
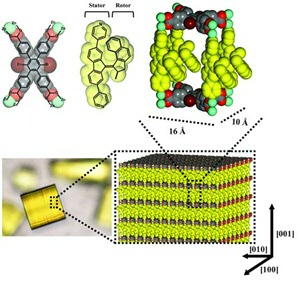
Motoren in een gel
Het werken met de eenvoudige moleculaire schakelaars is niet zonder problemen. Een daarvan is de Brownse beweging, de random beweging van deeltjes die ontstaat door botsing met snel bewegende moleculen uit de omgeving. Dit zorgt ervoor dat de zeer kleine motormoleculen voortdurend zonder richting rondtollen. Het is een hele uitdaging om de beweging toe controleren zodat ze bruikbaar zijn om werk te verzetten. Een manier om dat te doen is door schakelaars of motoren in een gel te stoppen, die de random bewegingen dempt. Een andere aanpak is het vastzetten van motormoleculen op een oppervlak. Op die manier blijken motoren en schakelaars in staat om werk te verrichten in de zichtbare wereld. Een mooi voorbeeld is de gecoördineerde beweging van motormoleculen die in een kooi-achtig materiaal zitten (een metaal-organisch frame) dat wellicht bruikbaar is om chemische reacties te katalyseren.
Kunstmatige spier
Een andere, spectaculaire demonstratie van deze aanpak is een kunstmatige spier die het team van Feringa maakte. Zij lieten vezels ontstaan via zelfassemblage van door licht aangedreven motormoleculen. Het licht kan niet doordringen tot in de vezel, dus wanneer slechts één kant werd beschenen ging de vezel buigen. Een onderzoek dat in 2018 werd gepubliceerd laat zien dat vezels van een hydrogel die zich kunnen buigen als spiervezels in staat zijn een gewicht van 0,4 milligram op te tillen. Daarmee is aangetoond dat moleculaire motoren werk kunnen doen in de gewone wereld. Dit maakt moleculaire machines ook interessant voor toepassingen in de elektronica. Er zijn inmiddels ook schakelaars ontwikkeld die niet alleen gevoelig zijn voor licht, maar ook voor zuur. Een recente studie, onder leiding van RUG-onderzoeker Ryan Chiechi, laat zien dat deze schakelaars reversibel kunnen worden vastgezet, zodat je er geheugenunits van kunt maken. Hoewel dit onderzoek nog in een erg pril stadium zit zou het op termijn kunnen bijdragen aan logische schakelingen en geheugen van elektronica.

Lichtgevoelige schakelaar
De motoren en schakelaars kunnen dus nuttig zijn in materiaalonderzoek en elektronica. Maar ze zijn ook van belang voor de biologie, aangezien de schakelaars de vorm van biologisch actieve moleculen kunnen veranderen, en daarmee ook hun functie. Het werk aan lichtgevoelige schakelaars in biologie en geneeskunde kwam rond 2011 echt op stoom. Veel werk op dit terrein is gedaan in samenwerking met Wiktor Szymanski, een medewerker van Feringa die zijn eigen onderzoeksgroep is begonnen in het UMCG. In 2013 werd een baanbrekend onderzoek gepubliceerd waarin de activiteit van een antibioticum is gecontroleerd met behulp van een lichtgevoelige schakelaar. Een overmaat aan antibiotica kan het ontstaan van resistentie veroorzaken. Willem Velema, promovendus van Feringa en Szymanski, kwam met het idee om antibiotica te maken die niet werken buiten het gebied waar ze nodig zijn. Hij maakte een schakelbaar antibioticum dat alleen na activatie met UV licht bacteriën kan doden. Zonder de belichting gaat de schakelaar weer in de ‘uit’ stand en is het antibioticum niet langer actief.
Maar de schakelaars hadden nog wel flinke beperkingen. Ze bestonden deels uit nogal grote, waterafstotende delen, waardoor ze niet goed oplosbaar waren. Bovendien reageerden ze alleen op UV licht, dat schadelijk is voor materialen en weefsels en bovendien niet ver in het lichaam doordringt. Een doorbraak kwam in 2017, in een onderzoek dat liet zien hoe schakelaars gevoelig zijn te maken voor infrarood licht. Dit type licht is niet schadelijk en dringt diep door in weefsels. Zo’n schakelaar is vervolgens ingebouwd in een antibioticum, trimethoprim. Als deze constructie met infrarood licht werd beschenen was er binnen twee uur een sterke toename te zien in de antibacteriële activiteit. Die schakeltijd is nog te lang om praktisch bruikbaar te zijn, dus er is nog meer ontwikkelwerk te doen.
Geneesmiddelen tegen kanker
Een vergelijkbaar route is gebruikt voor het maken van schakelbare geneesmiddelen tegen kanker. In 2019 presenteerde de Szymanski-groep een onderzoek naar remmers van kinasen, een klasse enzymen die betrokken is bij ongeveer de helft van alle vormen van uitgezaaide huidkanker (melanoom). Omdat kinasen van allerlei typen in grote hoeveelheden aanwezig zijn in veel verschillende weefsels is het moeilijk om een manier te vinden die heel selectief de schadelijke typen van deze enzymfamilie blokkeert. Daarom hebben kinaseremmers doorgaans veel bijwerkingen. Om dit probleem te omzeilen zijn kinaseremmers voorzien van een lichtgevoelige schakelaar. Op die manier is het mogelijk deze geneesmiddelen aan te zetten op de plek waar ze nodig zijn, in de tumor. Dat vermindert de effecten op gezond weefsel. Dit soort onderzoek moet leiden tot een behandeling die niet actief is in gezond weefsel en daar dus ook geen schade veroorzaakt.
Linker- en rechterhand
Tot slot weer terug naar de groep van Feringa zelf. In een van zijn meest recente publicaties bleek het mogelijk een speciale katalysator te maken, gebaseerd op een motormolecuul. Veel belangrijke moleculen, zoals geneesmiddelen, bestaan in twee chemisch identieke vormen die elkaars spiegelbeeld zijn, zoals een linker- en rechterhand. De twee ‘handen’ kunnen echter verschillende eigenschappen hebben: eentje is een geneesmiddel, terwijl de ander alleen bijwerkingen veroorzaakt. Met behulp van katalysatoren is het mogelijk gericht één van beide ‘handen’ te maken. De speciale katalysator van Feringa en postdoc Ruth Dorel is te schakelen tussen twee vormen die elk gericht één type ‘hand’ produceren.
Het is fascinerend om te zien hoe breed de toepassingsmogelijkheden van deze moleculen zijn in tal van velden. Al dit werk bouwt voort op de ontdekking die Feringa zijn Nobelprijs opleverde. Wel is het zo dat het onderzoek naar schakelbare geneesmiddelen en materialen nog in vroeg stadium van ontwikkeling zit. Er moet nog veel werk worden gedaan aan deze moleculaire motoren voordat ze op de markt komen.
Tekst: Antara Mazumdar & René Fransen
Met dank aan Wiktor Szymanski en Wojtek Danowski voor hun uitleg over recent onderzoek
Meer nieuws
-
19 mei 2026
Organiseer AI zoals het recht
