Ontdekking van Ben Feringa: Simpel proces levert complex polymeer op
Materialen van polymeren kunnen erg veelzijdig zijn, maar daar staat wel iets tegenover. Om complexe functionaliteit te krijgen, zoals zelf-reparerend vermogen, is doorgaans een ingewikkeld productieproces nodig. De uitdaging is dan ook om polymeren met slimme eigenschappen te maken via een eenvoudig proces. Dat is precies wat hoogleraar organische chemie Ben Feringa van de Rijksuniversiteit Groningen heeft gedaan, samen met collega-onderzoekers van de East China University of Science and Technology. In een artikel dat op 27 juli jl. verscheen in Science Advances beschrijven ze hoe een veelzijdig polymeer ontstaat via een simpel proces.

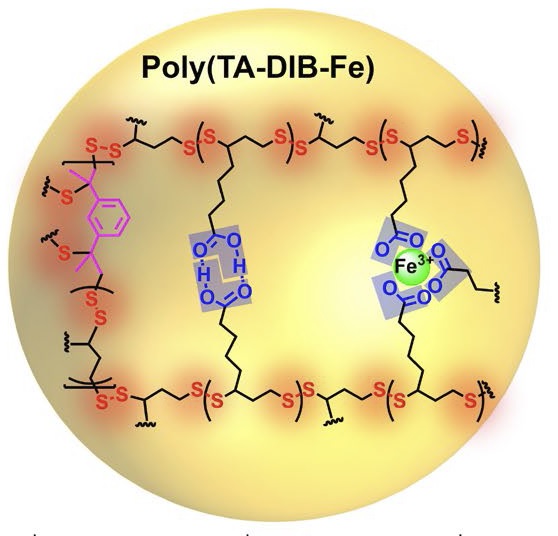
Zij begonnen met liponzuur, een natuurlijke molecuul. Door dit te smelten bij 70 graden Celsius en het daarna af te laten koelen vormt liponzuur een polymeer. Het probleem is dat dit materiaal niet zo stabiel is. Om daar iets aan te doen voegden Feringa en zijn collega’s de stoffen 1,3-diisopropenylbenzeen en ijzer(III) chloride toe tijdens het smelten van het liponzuur. Deze stoffen laten verbindingen in het polymeer ontstaan die zorgen voor stabiliteit.
Complex
Dit productieproces is heel simpel, maar het materiaal dat zo ontstaat heeft verschillende complexe eigenschappen. Het breekt niet onder druk en het is zelfs goed bestand tegen messen. Toch kan het polymeer tot wel 150 keer de oorspronkelijke lengte worden opgerekt zonder te breken. Krassen op het polymeer herstellen vanzelf bij kamertemperatuur. En als je slangetjes van het materiaal doorsnijdt en dan de snijvlakken tegen elkaar drukt vormen ze weer één geheel, met de sterkte van het originele materiaal.
Het polymeer kan zich ook verbinden aan oppervlakken door de vorming van waterstofbruggen, wat betekent dat het ook als lijm is te gebruiken. De onderzoekers smolten het polymeer en gebruikten de vloeistof om glazen plaatjes aan elkaar te lijmen. Een verlijmd oppervlak van 2 bij 2 centimeter kon een gewicht van vijf kilogram dragen. De auteurs concluderen dan ook dat ‘dit eenvoudige copolymeer belangrijke toepassingen kan hebben in de productie van lijm, zelf-reparerend materiaal en draagbare bio-afbreekbare producten.’
Feringa Nobel Prize Scientist Joint Research Center
Het werk dat in het artikel is beschreven, werd verricht in Shanghai, aan het Feringa Nobel Prize Scientist Joint Research Center, School of Chemistry and Molecular Engineering van de East China University of Science and Technology (ECUST). Prof. dr. Ben Feringa, die in november 2016 tot buitengewoon hoogleraar werd benoemd aan ECUST, bedacht en begeleidde het project met zijn Chinese collega’s. Het Feringa Nobel Prize Scientist Joint Research Center is in oktober 2017 geopend.
European Chemistry Gold Medal
Op 26 Augustus krijgt Ben Feringa de European Chemistry Gold Medal Award voor uitzonderlijke verdiensten in de scheikunde uitgereikt. Deze nieuwe prijs is in het leven geroepen door de European Chemical Society (EuChemS). ‘Not only is Ben a fantastic scientist, he is a great ambassador for the field of chemistry, a gifted communicator and a source of inspiration for many scientists in Europe and the rest of the world,’ schrijft EuChemS-bestuurslid Saskia van der Vies in de aankondiging.
Meer informatie
Referentie: Qi Zhang, Chen-Yu Shi, Da-Hui Qu, Yi-Tao Long, Ben L. Feringa and He Tian. Exploring a naturally tailored small molecule for stretchable, self-healing, and adhesive supramolecular polymers. Naar het volledige artikel.