Eerste chemisch aangedreven moleculaire motor

RUG-hoogleraar scheikunde Ben Feringa heeft een nieuwe primeur. Na de eerste moleculaire motor en de eerste moleculaire auto (allebei aangedreven door middel van licht) en nog een paar andere ontdekkingen heeft hij nu een ronddraaiend motormolecuul gemaakt dat loopt op chemische energie. De nieuwe motor is beschreven in Nature Chemistry van 6 juni.
Onze cellen zitten vol moleculaire motoren, de natuur weet heel goed hoe je die moet maken. Maar chemici en materiaalwetenschappers willen graag hun eigen motoren ontwikkelen, bijvoorbeeld om moleculaire machines mee aan te drijven. Ben Feringa is wereldwijd erkend als pionier in dit vakgebied. Hij heeft vooral moleculaire motortjes gemaakt die door licht worden aangedreven. Die zijn echter minder handig voor gebruik in het lichaam, of in een troebele oplossing.
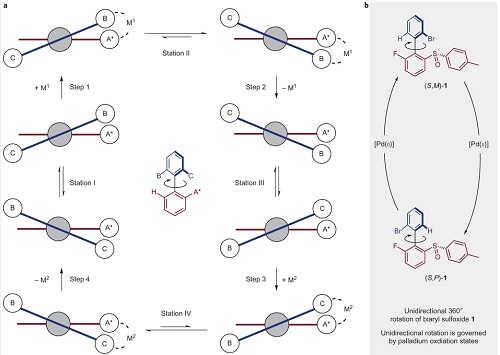
Mede daarom ging hij op zoek naar een manier om motortjes met een chemische aandrijving. Het moest opnieuw een draaiende rotor worden (zeg maar een scheepsschroef), gebaseerd op twee ringen waarvan er eentje kan ronddraaien, naar één kant. De twee ringen zijn met elkaar verbonden via twee koolstofatomen, die al draai-as dienen. Normaal gesproken blijft de bovenste ring onbeweeglijk staan door zwakke binding tussen atomen in het molecuul.
Feringa en zijn medewerkers bedachten dat een metaal als katalysator die binding kan losmaken, zodat het molecuul van vorm verandert en de bovenste ring een halve draai maakt. Daarna zou een tweede katalysator de ring nog een ‘duw’ moeten geven om een volledige omwenteling te produceren.
Wat Feringa toen bedacht is dat één katalysator wellicht beide stappen kan verzorgen. Een katalysator op basis van het metaal palladium kan de ring eerst oxideren en dan weer reduceren, wat de twee benodigde duwtjes zou opleveren. En het lukte inderdaad om de reactieomstandigheden waaronder dit kon plaatsvinden. De Groningse chemici lieten zien dat het werkte, maar het ging niet zo snel: in eerste instantie was er ongeveer een dag nodig om een verzameling motormoleculen in oplossing een volledige omwenteling te laten maken. Bovendien was er tussendoor een aanpassing van de temperatuur nodig en moesten nieuwe reagentia worden toegevoegd.
Maar vervolgens is de reactie gestroomlijnd waardoor deze sneller en eenvoudiger verliep, al bleek dat het aantal moleculen dat een volledige omwentelingen maakte hierdoor wat lager kwam te liggen. Dat was echter niet erg, dit experiment bewijst dat het mogelijk is om een moleculaire motor te maken die, aangedreven door chemische energie, een omwenteling in één richting maakt. En ook de door licht aangedreven moleculaire motor van Feringa is ooit zo begonnen: traag en complex, maar nu draait die op hoge snelheid rond. Wordt vervolgd dus…
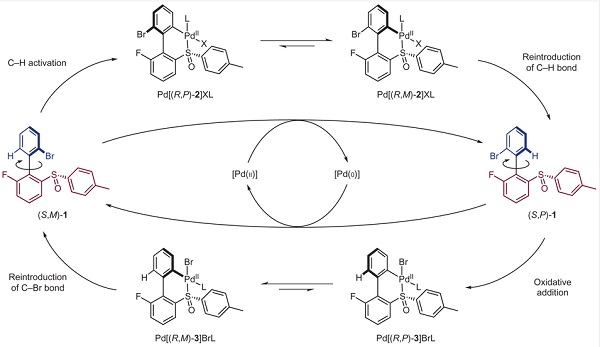
Referentie: Beatrice S. L. Collins, Jos C. M. Kistemaker, Edwin Otten & Ben L. Feringa, A chemically powered unidirectional rotary molecular motor based on a palladium redox cycle. Nature Chemistry, 6 June 2016, DOI 10.1038/nchem.2543
Zie ook dit bericht in De Volkskrant.
Tekst: René Fransen, ScienceLinx
Meer nieuws
-
28 april 2026
Hoe torenvalken turbulentie trotseren
