Recent Research Highlights
no longer maintained
see https://www.brownegroup.eu/
Identification of a rare Ni(IV) oxido complex. Sandeep Padamati (PhD Student) toegther with Prof Marcel Swart (university of Girona) and Prof Moniek Tromp (UVA) have identified a highly unusual Ni(IV) oxido complex that is formed upon reaction with bleach. The intermediate was identified spectroscopically and is active in C-H oxidation opening the possibility of taming the highly reactive oxidant NaOCl to do selective oxidations.
Padamati, S. K., Angelone, D., Draksharapu, A., Primi, G., Martin, D. J., Tromp, M., Swart M., Browne W.R., Transient Formation and Reactivity of a High-Valent Nickel(IV) Oxido Complex., J. Am. Chem. Soc. 2017 139, 8718-8724. DOI: 10.1021/jacs.7b04158
Fully reversible inorganic photochromic switch. Duenpen Unjaroen (PhD student) together with Hans Kasper (Bachelor research student) have developed a fully reversible photochromic switch which is based solely on metal centred photochemistry - i.e. reversible photoinduced ligand dissociation. This new switch is based on a ruthenium(II) complex of a ligand with a long history in Groningen - MeN4Py (1,1-di(pyridin-2-yl)-N,N’-bis(pyridin-2-yl-methyl)-ethan-1-amine) and is better known as a ligand for iron(II) based oxidation catalysis.
Duenpen Unjaroen, Johan B. Kasper and W. R. Browne* "Reversible photochromic switching in a Ru(II) polypyridyl complex" Dalton, 2014, 43, 16974-16976. http://dx.doi.org/10.1039/C4DT02430C
________________________________________________________________________________________________________________________
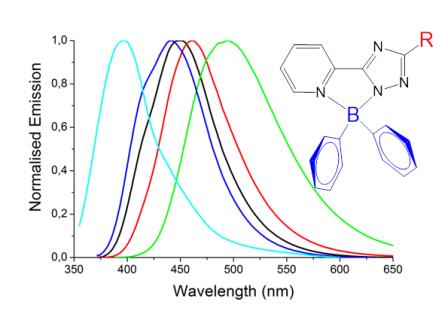
New high quantum yield blue emitter s for application in Liquid Scintillator Technologies. In an upcoming paper, the development of a new class of blue emitters based on cheap and scalable syntheses of boron-1,2,4-triazoles will be described. These fluorophores are being evaulated for use in liquid scintillators for radiation detection as part of a cooperation between the Browne group and the team at INCAS3 lead by Dr Heinrich Wörtche.
P. Dijkstra, Davide Angelone, E. Taknishnikh, H. J. Wörtche, E. Otten, W. R. Browne*, "Pyridyl-1,2,4-triazole diphenyl boron complexes as efficient tuneable blue emitters" Dalton, 2014, 43, 17740 - 17745, http://dx.doi.org/10.1039/C4DT01393
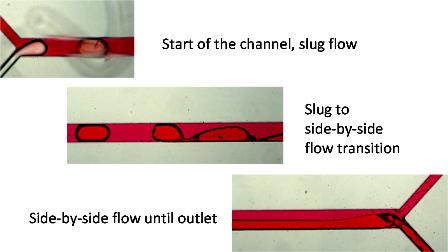
Controlling flow profiles in a microfluidic channel is a major research challenge. In a paper to Lab on a Chip by Hella Logtenberg (Browne research group) and Maria Lopez-Martinez (Verpoorte research group) controlled switching between slug-flow and side-by-side flow in a a single channel is obtained by a deceptively simple surface modification strategy that employs spatially controlled polymer coating of channel surfaces.
"Multiple flow profiles for two-phase flow in single microfluidic channels through site-selective channel coating" Lab on a Chip, 2011, 11, 2030-2034. http://dx.doi.org/10.1039/C1LC20012G
A major challenge however is to coat channel surfaces with responsive materials such as redox polymers. Recently we reported on the insitu electropolymerisation of a sexithiophene polymer and demonstrated the challenges face in in situ characterisation and the opportunities provided by resonance Raman spectroscopy.
"In situ monitoring of polymer redox states by Resonance microRaman spectroscopy and its applications in polymer modified microfluidic channels" Analytical Methods, 2012, 4, 73-79. http://dx.doi.org/10.1039/C1AY05475A
Oxidation Catalysis
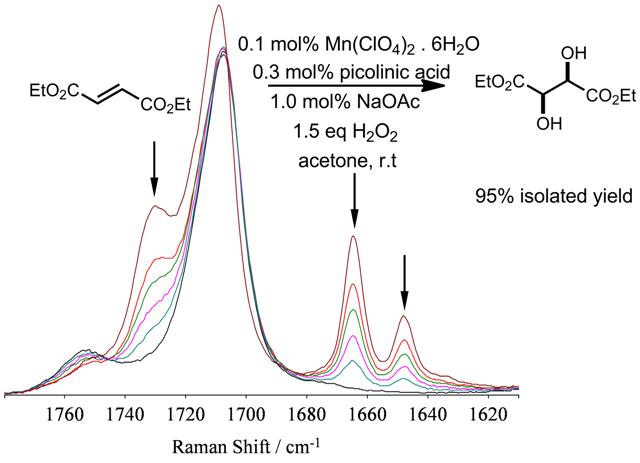
In two recent articles in Dalton and Organic & Biomolecular Chemistry, a new remarkably simple method for the, amongst other oxidative transformations, cis-dihydroxylation of electron deficient alkenes with H2O2 is described. The method uses a simple combination of catalytic amounts (0.1-1 mol%) of a manganese salt, picolinic acid, a base in acetone with near stoichiometric amounts of H2O2 in acetone. Several examples showing complete conversion with near quantitative yield of the cis-diol product are reported.
The discovery and development of this system has relied heavily on the use of Raman spectroscopy for reaction monitoring.
The project was performed by Dr Dirk Pijper and Pattama Saisaha together with Dr Ruben van Summeren and Dr Paul Alsters at DSM. The discovery of this new system has revealed that many early systems based on picolyl based ligands actually work by first ligand oxidation to picolinic acid.
"Manganese Catalyzed cis-Dihydroxylation of Electron Deficient Alkenes with H2O2" Org. Biomol. Chem., 2010, 8, 4444-4450. http://dx.doi.org/10.1039/c0ob00102c
W. R. Browne, P. L. Alsters, R. P. van Summeren, E. Ijpeij, J. W. de Boer, P. Saisaha, D. Pijper, B. L. Feringa, Composition for oxidation of Organic substrates, PCT Int. Appl. (2011), WO 2011104333