The evolutionary ecology of a mutualism
Mutualisms, interactions in which individuals of different species cooperate, are wide-spread throughout nature. Examples include plants and their pollinators, cleaner fish and their clients and light-producing bacteria that inhabit bobtail squids. In fact, in the form of mitochondria, mutualism stood at the basis of life as we now it. Recently, mutualisms also are increasingly recognized for their importance to ecosystem diversity and stability. Consequently, an increasing number of studies aims to unravel details of specific interactions such as the natural history of the mutualisms, costs and benefits for the partner species involved and the effects of these mutualisms on their ecosystem.
Despite this increasing focus on mutualisms, their evolution and evolutionary stability are still poorly understood. How does cooperation between different species come off the ground? And once a mutualistic interaction has been established, how does it stay stable over evolutionary time? Where two species live this closely together, the interests of the partner species are expected to never be fully aligned, potentially leading to conflicts disrupting the interaction. The many stable mutualisms we see in nature are therefore possibly results of ongoing mutual exploitation by the partners with net benefits for both, rather than harmonious cooperation. In our project, we aim to shed light on these questions using a combined theoretical and empirical approach. Here we will focus on the empirical part of the project.
Cultivation mutualisms are mutualisms in which a host species promotes the growth of the symbiont species which it consumes or on which it relies for food. Classical examples of cultivation mutualisms are algae-growing damselfish and fungus-growing ants. Both ants and damselfish actively grow their symbiont species as a crop for food in especially constructed gardens. Similarly, root-aphid tending ants keep aphids as cattle in aphid-chambers in their nest. The ants rely for their sugar consumption on the honeydew the aphids produce and in return the ants provide the aphids with housing and active protection against predators. It is the latter system of aphid tending ants that is the focal system of our research. Like other mutualisms, also cultivation mutualism can be prone to destabilizing conflicts of interests between the partner species. Three major potential sources of conflict are energy allocation of the symbiont towards sexual reproduction, competition with other symbionts (due to high within-host symbiont diversity) and dispersal. This energy allocation may compromise symbiont productivity and it would therefore be in the host’s interest if the symbiont invested all energy in productivity. However, the symbiont might benefit from sexually reproducing, outcompeting other symbionts or dispersing and might prefer to allocate at least some energy to these. How are these conflicts resolved in an aphid-ant mutualism? What mode of reproduction do the symbiont aphids have? What is their degree of diversity (and thus competition) and to what extent do they disperse independently of the ants?
We investigated these questions in a particularly interesting cultivation mutualism: the interaction of the yellow meadow ant Lasius flavus and the root-aphids it tends in its nest. More than ten different species of root-aphids have been found in L. flavus nests, where they feed on the roots of various grasses. The ants are said to be dependent on the honeydew produced by these aphids as their sole sugar source and are also known to occasionally prey on the aphids. We conducted a population genetic study on these aphids, focusing on the four most common and abundant species: Geoica utricularia, Tetraneura ulmi, Forda marginata and its sibling species Forda formicaria. To this end, we sampled aphids from ant mounds along a transect on the island of Schiermonnikoog. About 500 aphids were collected and genotyped with species-specific sets of microsatellite markers, developed by us prior to this study.
The results show that all four focal aphid species reproduce predominantly clonally in the sampled population. Year-round clonal reproduction had been described before for G. utricularia and both Forda species, but it was surprising that also T. ulmi had apparently lost sexual reproduction. In order to gain insight in aphid diversity and dispersal levels, we mapped the distribution of clonal lineages over the island and over and within ant mounds (Figure 1). All four species have a single predominant clone to which the majority of the collected aphids belongs. A couple of clones occur at several transect locations, but only one clonal lineage occurs along the whole transect. This indicates that aphids do occasionally disperse, but that this dispersal is very limited. Aphids did often occur in monocultures (in terms of species as well as clonal lineages) but not exclusively. The scope for between-aphid competition is further reduced by the aphid species and clonal lineages occurring spatially segregated in the ant mounds. Lastly, the same aphid clonal lineages seem to stick with the same ant mounds over time as no major turn over in aphid composition was observed over years.
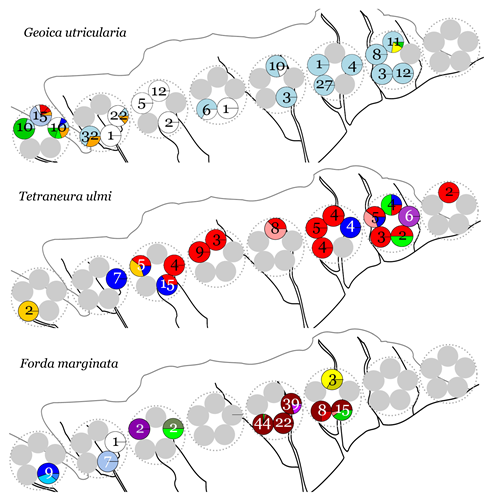
To conclude, in this system the aphid symbionts do not reproduce sexually, do occur at low degrees of (genetic) diversity and show low levels of dispersal. These symbiont characteristics show remarkable similarity to what is observed in fungus-growing ants. Also their symbiont fungus reproduces clonally, disperses independently to a limited extent and occurs as a monoculture in the garden. Whether we are dealing here with conflict resolutions universal for cultivation mutualisms will be further investigated in the theoretical part of this project.
