Sperm competition and strategic male mating effort: theoretical and experimental approaches
My research falls broadly in the field of evolutionary ecology. I am mainly interested in sperm competition and life history evolution. The focus of my work has been on strategic mating effort. When males have high costs of mating - they might provide females with nuptial gifts or need to produce a large ejaculate in order to be successful in sperm competition - they face a problem. As resources will be limited, they need to allocate their mating investment over successive matings. The focus of my work has been to understand how high costs of mating and the resulting trade-off between present and future mating effort will select for prudent male mating strategies. During the last years I have integrated theoretical and experimental work using Panorpa scorpionflies as model organisms to increase our understanding of strategic mating effort. Below I review some of the recent research highlights.
Male mating effort in scorpionflies
The mating systems of scorpionflies are characterised by the adoption of different male mating tactics. In general, male scorpionflies may use one of three different mating tactics: they may either offer females a) a dead arthropod (a prey) or b) a salivary secretion, which are consumed by the female during copulation, or c) they may attempt to mate with the female without nuptial food gifts. However in P. cognata, my main study object, matings are always accompanied by nuptial food gifts. The most beneficial tactic, but also the most costly, is the offering of salivary masses. Females feed on the salivary mass during copulation, and terminate copulation when they have consumed it (Fig. 16). Saliva production is costly, prompting the question of the benefit to males of producing these gifts.


One line of research has therefore focused on the function of these nuptial gifts. We have been able to demonstrate that the main function is mating effort. Potentially, nuptial gifts might also function as paternal effort. Female scorpionflies (and their offspring) gain some nutritional benefits from nuptial gift consumption, but the effect is marginal. On the other hand, there is a threefold function as mating effort. First, at least in P. cognata, males must present a nuptial gift in order to achieve a copulation. Second, the production of larger nuptial gifts lead to copulations of longer duration, which increase female sperm storage as sperm transfer is continuous during copulation. By using molecular techniques we have studied the mechanism of sperm competition in scorpionflies and concluded that it largely conforms to a "fair raffle" of sperm (the numerical representation of sperm influences the fertilization probability). Thus ultimately, by producing a larger salivary mass, males increase fertilization success. We have also shown that by offering females a large salivary, males increase the female refractory period following mating and thus avoid or decrease the level of future sperm competition.
Strategic mating effort
Males thus increase their reproductive success in a current mating situation by producing a large salivary mass. However, saliva production is strongly dependent on male food intake and many males have a very restricted amount of saliva in their salivary glands. Therefore, males have to conserve some resources for future matings (potentially with higher quality females). This trade-off between present and future male mating investment will be expected to select for strategic mating effort. One would expect males to increase investment in matings, where the benefit (in terms of sired offspring per investment) will be higher. Two factors have been shown to be especially important: female quality and the intensity of sperm competition. To study the influence of female quality on male mating decisions, we compared the size of the salivary masses produced by males in matings with high and low quality females, where quality is determined by female egg load. We found that males are able to discriminate between females of different fecundity and offer larger salivary masses in matings with high quality females, an example of cryptic male choice (Fig. 17).
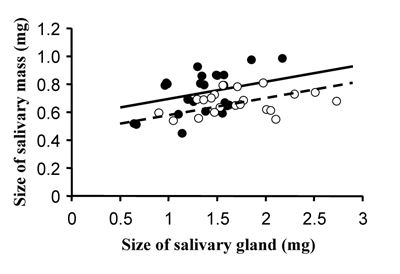

Female scorpionflies usually mate with many males. Thus often females already have stored sperm from rival males when males mate with them. If female vary in the number of sperm previously stored, the expected gain, in terms of gained fertilizations per unit male mating investment will differ between matings, and as a result males are expected to invest strategically. If males are certain of sperm competition, they should respond to an increased intensity of sperm competition by investing less in a mating. The cause for this somewhat counterintuitive prediction is that the marginal fitness increase per each additional investment will be lower the more rival sperm is competing. In analogy with a lottery, the value of buying extra tickets in a raffle for a fixed prize becomes greater the fewer the competitors. Thus, it should pay males to invest less in raffles with high sperm competition intensity.
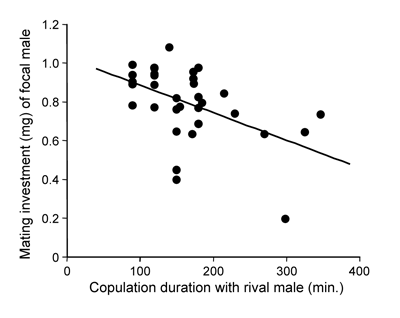

To test this prediction in scorpionflies, we measured male mating investment in response to the number of foreign sperm stored by females in a previous mating. Female sperm storage was manipulated by interrupting copulations at different time points. This experiment demonstrated that resource-limited males invest strategically in relation to the amount of sperm stored by females in a previous mating. We found continuously decreasing male investment (measured as the size of the salivary mass) in response to increasing amounts of competing sperm (Fig. 18). These results demonstrate an unprecedented male ability to assess the number of sperm stored by females. As a result, males are capable of an extraordinarily fine-tuned reaction to the intensity of sperm competition.
Theoretical analyses of sperm allocation in relation to female mating status
The theoretical framework of strategic mating effort comes from life history and sperm competition theory. Previous theoretical models have predicted that males should allocate more sperm in matings where the risk of sperm competition is high. The logic behind this argument is quite straightforward. If the risk of sperm competition is low, males should invest less because he will fertilize most female eggs anyway irrespective of investment. It has therefore often been argued that males should invest less sperm in matings with virgin females compared to matings with already mated females, because non-mated females represent a lower risk of sperm competition. However, in species where females mate very frequently, high sperm competition risk will covary with high sperm competition intensity leading to more unpredictable conditions, as high competition intensity should favour smaller ejaculates. Furthermore, empirical studies on sperm expenditure in relation to female mating status have thus far produced very disparate results.
We therefore performed a theoretical analysis of how males should respond to variation in female mating status (e.g. virgin vs. mated) in mating systems with internal fertilization. In our simulations, we found that males should allocate more sperm in matings with virgin females when female mating frequency is relatively high, whereas low remating rates will select for higher effort in matings with nonvirgin females. (Fig. 19).
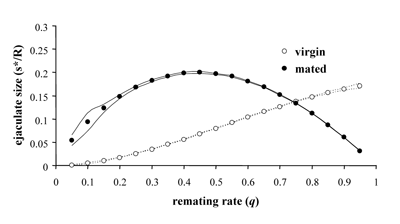

These results shed some light on several findings that have been difficult to explain adaptively by the hitherto developed theory on sperm allocation. Some studies do indeed support the predominant view that males should allocate most sperm to matings with nonvirgin females. However, the opposite case, higher ejaculate expenditure in matings with virgin females, has been found in studies from a wide diversity of species, including mites, orb-weaving spiders, bush crickets, bedbugs and stalk-eyed flies. Hitherto, these data have been difficult to explain, but the predictions from our model might allow interpreting these data from the perspective of optimal male sperm allocation. Furthermore, these novel predictions have already inspired new empirical work in this exciting research field.
