Polymer Chemistry and Bioengineering
Disclaimer: Prof. Dr. Herrmann moved to DWI Leibniz-Institute for Interactive Materials and Aachen University. He maintains a honorary position at ZIAM, but is no longer recruiting students at RUG.
Our group deals with engineered biomacromolecules or bioorganic hybrid structures for various medical and technological applications. These materials are fabricated by integrating tools from Organic- and Polymer Chemistry as well as from Molecular Biology. For evolving complex functions the accurate assembly of different building blocks into well-defined nanoscopic aggregates is an important requirement. For their characterization various nanotechnology tools like scanning force microscopy, transmission electron microscopy and fluorescence-based techniques are utilized. Finally, the interactions of the assemblies with living matter are explored, e.g. in the context of advanced drug delivery vehicles. Other applications are diagnostic and nano-electronic devices or the bottom-up construction of proto-cells.
DNA-Polymer Hybrid Materials
We develop chemical and molecular biology methods to prepare DNA block copolymers (DBCs) that consist of an oligonucleotide segment that is covalently connected to a synthetic polymer unit. DBCs with a hydrophobic polymer microphase separate and self-assemble into aggregates with sizes in the nanometer to micrometer range. One of them is DBC micelles, which consist of a core containing the hydrophobic polymer while the corona is constituted of oligonucleotides. These nanoobjects were successfully employed for drug delivery (Figure 1). Thereby, the single-stranded DNA at the rim was equipped with folate targeting units to achieve internalization into cancerous cells by a very simple hybridization procedure. In the interior of the DNA nanocarriers a hydrophobic anticancer drug was introduced. If both, the targeting units and the drug, act in concert cancerous cells were efficiently killed.
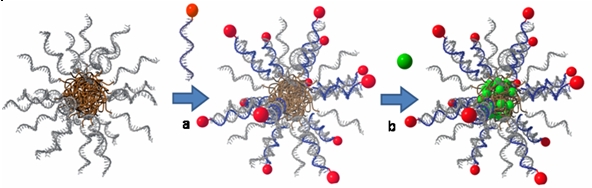
These experiments demonstrated that DNA block copolymer aggregates are a platform allowing for combinatorial testing of drug carrier systems. Many combinations of targeting and diagnostics units together with different drugs can be screened in short time due to the ease of functionalization of the nanoobjects.
de Vries JW, Zhang F, Herrmann A: Drug Delivery Systems Based on Nucleic Acid Nanostructures. J. Control. Release 2013, 172(2): 467-483.
Kwak M, Herrmann A: Nucleic Acid/Organic Polymer Hybrid Materials: Synthesis, Superstructures, and Applications. Angew. Chem. Int. Ed. 2010, 49(46): 8574-8587.
Supramolecular Protective Groups
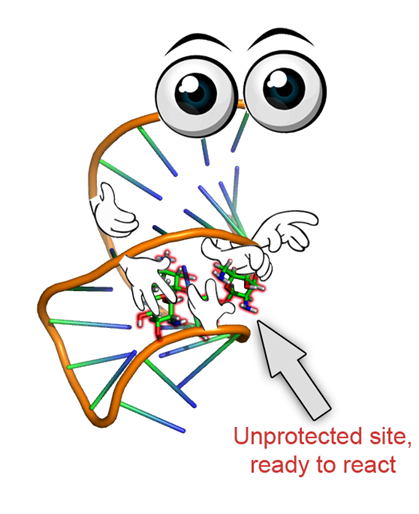
Besides exploring DBCs where nucleic acids are connected via covalent bonds we exploit supramolecular interactions between aptamers and complex natural products. Natural products are frequently used as active drug molecules but very often they need to be site-specifically modified to achieve lower toxicity, higher stability in the body or better pharmacokinetics. Usually, this requires cumbersome total synthesis including many reaction steps. Our group developed an intriguing way around this dilemma. With neomycin B as a natural antiobiotic containing 7 hydroxyl groups and 6 amine groups with very similar chemical reactivity we demonstrated regioselective functionalization within only a single synthetic step. Therefore, at the stage of the natural product we introduced aptamers that bind several functionalities of the molecule while other functional groups not in contact with the aptamer can be modified selectively. In this way, many new antibiotic derivatives were fabricated that would have required years to be synthesized by conventional procedures. For that reason the aptamer protective group technology will have great impact on natural product-based drug development and lead optimization.
Bastian AA, Marcozzi A, Herrmann A: Selective Transformations of Complex Molecules are Enabled by Aptameric Protective Groups. Nat. Chem. 2012, 4: 789–793.
Supercharged Polypeptides
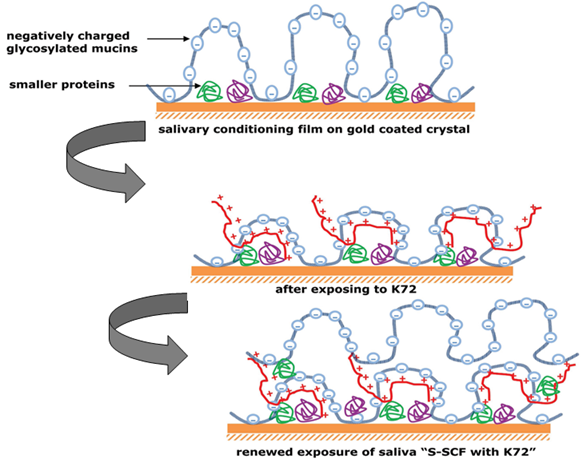
Our group pioneered non-folded supercharged polypeptides (SUPs). As a scaffold we selected elastin-like proteins that consist of a repetitive pentapeptide sequence. One of the five neutral amino acids can be replaced by either glutamic acid or lysine allowing to produce highly negatively and positively charged peptide polymers, respectively. The resulting biological polyelectrolytes were employed to fabricate superstructures by the layer-by-layer method thereby exploiting the electrostatic interactions of oppositely charged variants. One example is hollow capsules for drug delivery. Another example represents positively charged SUPs like K72, which was used to stabilize saliva conditioning films to improve the mastication and speech of patients with impaired oral biolubrication.
Veeregowda DH, Kolbe A, van der Mei HC, Busscher HJ, Herrmann A, Sharma PK:
Recombinant Supercharged Polypeptides Restore and Improve Biolubrication.
Adv. Mater. 2013, 25(25): 3426-3431.
Kolbe A, del Mercato LL, Abbasi AZ, Rivera Gil P, Gorzini SJ, Huibers WHC, Poolman B, Parak WJ, Herrmann A:
De Novo Design of Supercharged, Unfolded Protein Polymers, and Their Assembly into Supramolecular Aggregates.
Macromol. Rapid Commun. 2011, 32(2): 186-190.
| Last modified: | 21 February 2019 3.33 p.m. |
