
Department of Experimental Hematology

The general aims of the division of Experimental Hematology are to obtain detailed knowledge on (molecular) mechanisms that determine hematopoietic stem cell self-renewal and differentiation, with the ultimate goal to deepen our insights in the development of human leukemias. We have a specific interest in dissecting the clonal heterogeneity of leukemias and recently developed tools that allow us to identify and prospectively isolate genetically distinct clones within individual patients. We perform gene-function analyses in human hematopoietic stem and progenitor cells isolated from cordblood and bone marrow utilizing various strategies including optimized retro/lentiviral transduction protocols and inducible RNAi and CRISPR/Cas9 approaches. We have a longstanding expertise in using molecular approaches (transcriptome, proteome and epigenome) to further understand processes such as hematopoietic differentiation, proliferation, apoptosis and self-renewal. We utilize a series of cell biological in vitro and in vivo model systems to be able to translate knowledge from our molecular research lines to more general concepts of hematopoietic stem cell biology. We make use of sophisticated humanize niche xenograft models in order to mimic that situation in leukemia patients as closely as possible. Furthermore, we directly collaborate with the clinical and diagnostic departments at the UMCG and have banked large series of leukemic patient material and are currently focusing on elucidating whether leukemic stem cells exist, what their phenotype is, which mechanisms result in enhanced self-renewal and a disturbed differentiation profile, and whether we can restore the normal hematopoietic differentiation program by e.g. interfering at the genetic level or by utilizing novel therapeutical agents. We anticipate that our studies will lead to a more rational approach in the clinic of this highly malignant disorder.
LATEST NEWS!!
VACANCY: we are looking for a motivated technician on a project focussing on the epigenome of leukemic stem cells.
For info, please contact JJ Schuringa (j.j.schuringa@umcg.nl, 0031-50-3619391)
to apply: https://noorderlink.nl/participanten/umc-groningen/10419-02S0005FNP
.......
October 2019
Le Duy Minh starts as PhD student on his awarded MPDI proposal Comprehensively targeting acute myeloid leukemia with CAR T cells. Welcome!
September 2019
Laura Dillingh starts as PhD student on a network grant awarded by the ZIAM/RUG entitled The Physics of Cancer. Laura's project is a collaborative effort with the labs of Jan Jacob Schuringa, Wouter Roos and George Palasantzas to unravel physical properties of normal hematopoietic stem cells and leukemic stem cells using AFM. For further info, see phycan.nl. Looking forward to this exciting collaboration!

June 2019
Vincent van den Boom publishes his work on epigenetic instability as a consequence of cellular stress in eLIFE. Nice collaboration with the Cell Biology Department (Harrie Kampinga and Steven Bergink labs).

June 2019
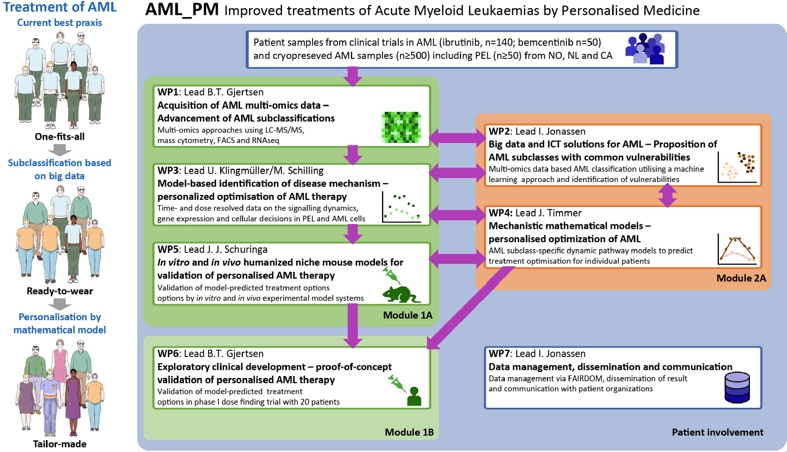
Emmanuel Griessinger starts as postdoc in the lab. He will work on an EU EraPerMed network which aims to unravel the molecular biology of erythoid leukemias and AML focussed on personalized medicine. Great collaborative initiative with the University of Bergen (Bjorn Gjertsen, Inge Jonassen), University of Heidelberg (Ursula Klingmuller, Marcel Schilling), University of Freiburg (Jens Timmer) and University Medical Center Groningen/University of Groningen (JJ Schuringa).
June 2019
Isabel Weinhauser recieves UMCG/FAPESP PhD fellowship for her work on APL and AML on the role of tumor supportive vs tumor suppressive macrophages. Well done Isabel!
June 2019
Mylene Gerritsen publishes her work on RUNX1 mutations in AML in Blood Advances.

May 2019
Our consortium Physics of Cancer: Cancer as a Materials Science problem was awarded by the ZIAM. For details see our website. The aim of this current program is to study the stiffness of cells and tissues for two partially opposing types of cancer by means of careful experiments and multiscale computational modelling. Subsequently, we aim to identify key malfunctioning proteins inside cells, on the cell membrane as well as in the extracellular matrix, by a combination of computational modelling, structure analysis and mutation studies. Interference with these proteins could become a future target to prevent the release of cancer cells from a tumour, invasion and further development of the tumour. Five PhD students will embark on this ambitous project. Great collaboration between the ZIAM/RUG and UMCG. Laura Dillingh will start early September as one of the PhD students!
January 2019
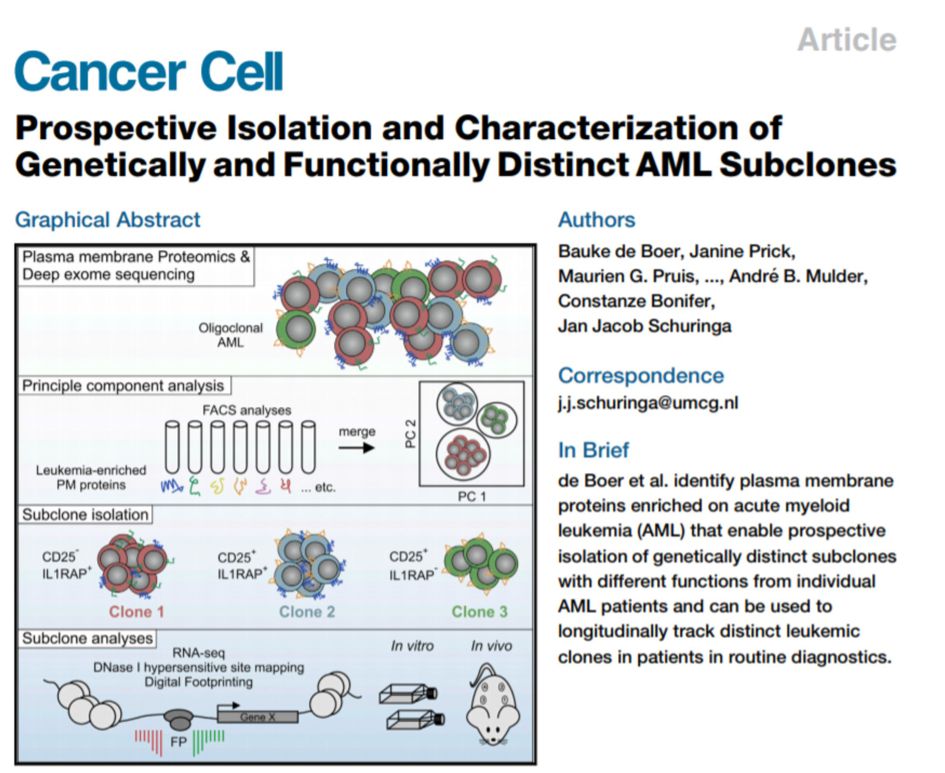
Bauke de Boer wins the annual award of Dutch TOP publication at the Dutch Hematology Congress for his presentation on his Cancer Cell paper Prospective isolation and Characterization of Genetically and Functionally Distinct Subclones in AML. Well done Bauke!

January 2019
The Joost Martens lab (Nijmegen) publishes their work on epigenomes in AML in Cell Reports. Great to have been part of this, well done Joost!
October 2018
ERA PerMed grant of 1.5M euro awarded to JJ Schuringa and partners in Bergen (Norway), Heidelberg and Freiburg (Germany). Within this proposal we will employ multi-omics technologies at the single cell level and perform systems biology modeling in order to unravel derailed signaling transduction in AML to be able to design personalized treatment strategies.
Vacancy for a 3-year postdoc position!
October 2018
paper by Bauke de Boer et al published in Cancer Cell, including issue highlight and Editorial
------------------------------------------------------
August 2018
What is Leukemia? explained to the lay audience (in Dutch)


--------------------------------------
July 2018
KWF project entitled Dissecting clonal heterogeneity in AML using the plasma membrane proteome was selected by Maarten van der Weijden as one of the 11 fundraising projects in his heroic effort to swim 200 km in the elfstedenzwemtocht! FANTASTIC!
see also:
https://www.11stedenzwemtocht.nl/onderzoek/
https://www.kwf.nl/onderzoek/welk-onderzoek-krijgt-geld/Pages/Elfstedenzwemtocht.aspx

--------------------------------------
July 2018
Our time to organize the workshop for our ITN HaemMetabolome in Groningen. Great meeting, and great sailing afterwards at the Paterswoldse meer!

--------------------------------------
July 2018
Susan Hilgendorf defends her thesis entitled The Long road: The autophagic network and TP53/ASXL1 aberrations in hematopoietic malignancies.

--------------------------------------
December 2017
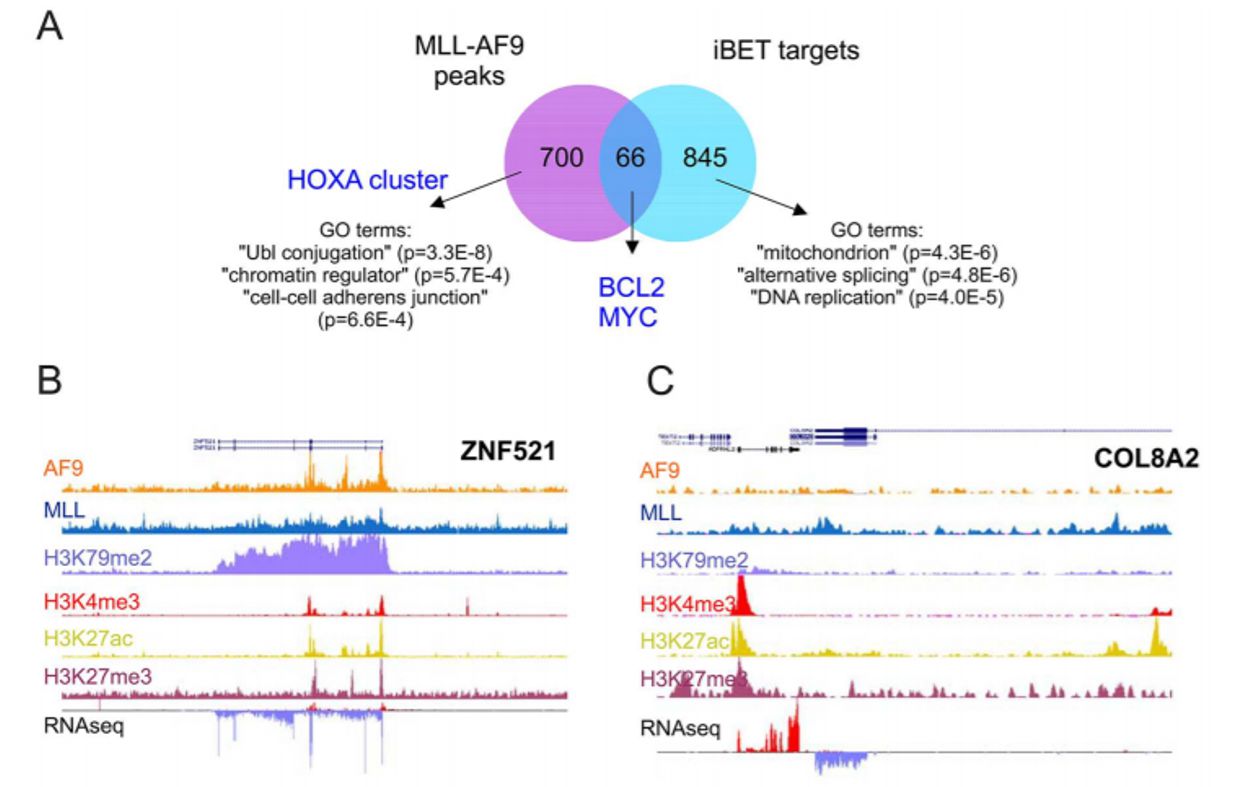
We performed transcriptome and ChIPseq experiments to compare direct and indirect targets of MLL-AF9. We conclude that BRD3/4 and the FLT3-TAK1/NF-kB pathways collectively control a set of targets that are critically important for the survival of human MLL-AF9 cells. Marco published his work in Plos ONE, which was a collaborative effort together with the Joost Martens lab in Nijmegen.
--------------------------------------
October 2017
JJ co-organized EMBO workshop in Heidelberg on Humanized Mice in Biomedical Research.
--------------------------------------
July 2017
Hendrik published his work on autophagy in leukemia, in Cell Death and Differentiation. The work indicates that targeting autophagy might provide new therapeutic options for treatment of AML
--------------------------------------
July 2017
Further improvement of the humanized niche xenograft model! We genetically engineered human MSCs to secrete human interleukin-3 (IL-3) and thrombopoietin (TPO). In vivo, these genetically engineered MSCs maintained their ability to differentiate into bone, adipocytes, and other stromal components. Upon transplantation of MLL-AF9-transduced CB CD34+ cells, acute myeloid leukemia (AML) and acute lymphoblastic leukemia (ALL) developed in engineered scaffolds, in which a significantly higher percentage of myeloid clones was observed in the mouse compartments compared with previous models. This approach will be useful for functional study of the importance of niche factors in normal and malignant human hematopoiesis. Work by Marco and Bauke, published in Experimental Hematology.

--------------------------------------
March 2017
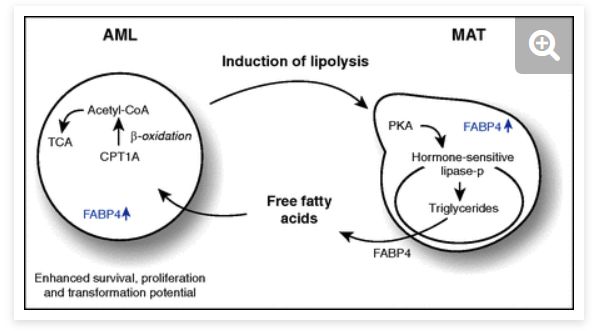
Smart niche usage: release its fat and burn it! JJ wrote an Inside Blood editorial on a paper by the Rushworth lab
--------------------------------------
June 2017
KWF grant awarded to JJ Schuringa entitled: Using the plasma membrane proteome to dissect clonal heterogeneity and track disease progression in AML. Two PhD positions available!! See Vacancies


--------------------------------------
October 2016
Antonella publishes her work on the development of improved humanized niche xenograft models. By implanting ceramic scaffolds coated with human mesenchymal stromal cells into immune-deficient mice, we were able to mimic the human bone marrow niche. Thus, we have established a human leukemia xenograft mouse model in which a large cohort of patient samples successfully engrafted, which covered all of the important genetic and risk subgroups. We found that by providing a humanized environment, stem cell self-renewal properties were better maintained as determined by serial transplantation assays and genome-wide transcriptome studies, and less clonal drift was observed as determined by exome sequencing. The human leukemia xenograft mouse models that we have established here will serve as an excellent resource for future studies aimed at exploring novel therapeutic approaches. The was a collaboration with Richard Groen, and was published in Blood. Also got the cover image and Inside Blood editorial.

--------------------------------------
October 2016
Pallavi and Marco published their work on BCR-ABL and MLL-AF9-induced leukemia making use of humanized niche xenograft models in Leukemia.

--------------------------------------
September 2016
Susan publishes her work on ASXL1 in Experimental Hematology. We conclude that ASXL1 is essential for erythroid development and differentiation and that the aberrant differentiation is, at least in part, facilitated via PRC2.

--------------------------------------
September 2016
Thesis defense Pallavi Sontakke: Extrinsic and intrinsic cues involved in BCR-ABL induced leukemogenesis
--------------------------------------
June 2016
Hendrik Folkerts published his nice work on autophagy in human CD34+ cells in Stem Cells


--------------------------------------
April 2016
Pallavi published our work on metabolism in human leukemias in collaboration with the Ulrich Gunther lab in Birmingham in Plos One. From this collaboration we later on developed the ITN HaemMetabolome consortium.

--------------------------------------
February 2016
We contributed to a European consensus document published in Haematologica

--------------------------------------
January 2016
Vincent van den Boom and Henny Maat published their work on the non-canonical PRC1.1 complex in Cell Reports. see website
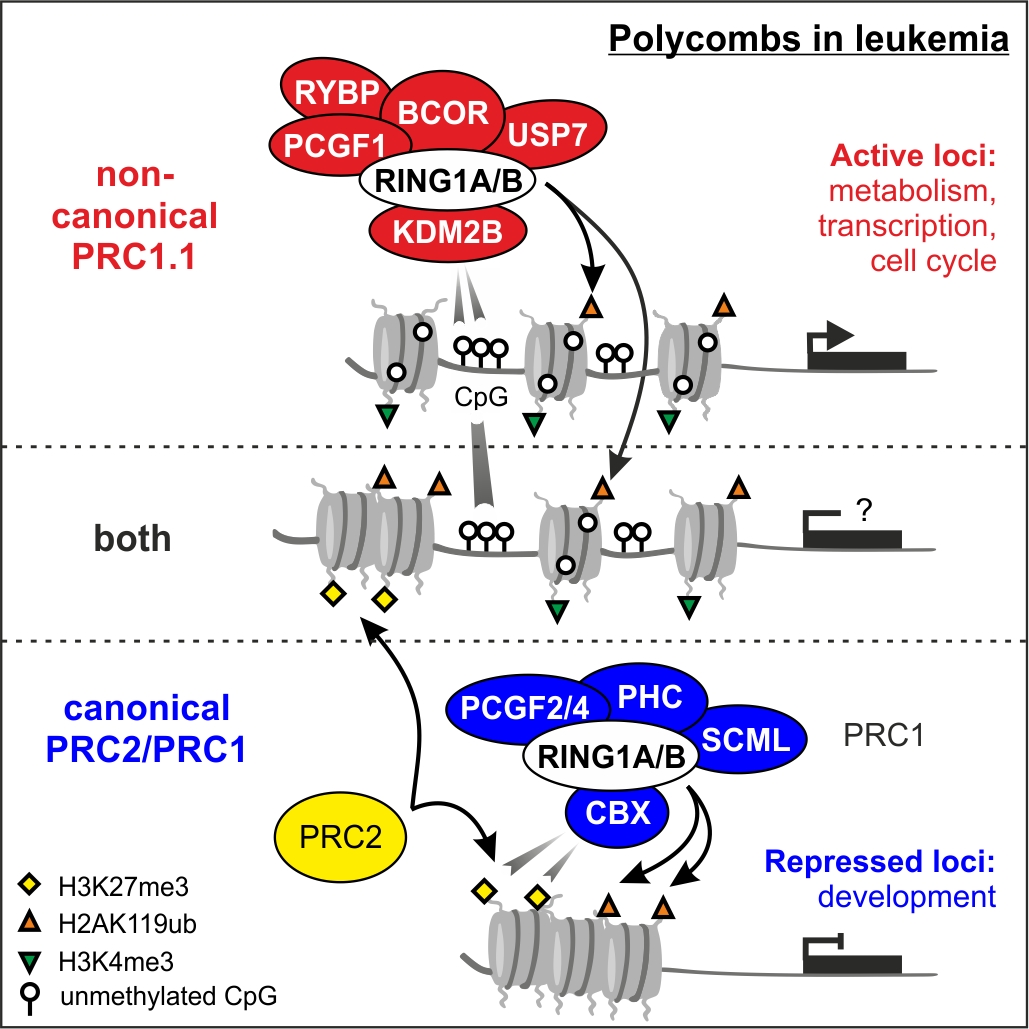

--------------------------------------
October 2015
Matthieu Bosman defended his thesis in October 2015.

--------------------------------------
September 2015
--------------------------------------
May 2015

Marta Capala published a paper in Plos ONE showing that RAC2 fulfils an essential role in BCR-ABL transformed cells. Time-lapse confocal microscopy surprisingly revealed very distinct localisation of RAC1 and RAC2 in human hematopoietic stem/progenitor cells. Proteome studies to identify RAC1 and RAC2 interactomes revealed that RAC2 interacts with a set of mitochondrial proteins including mitochondrial transport proteins SAM50 and Metaxin 1. Downregulation of RAC2 or SAM50 impaired self-renewal associated with a decreased mitochondrial membrane potential and impaired mitochondrial integrity as determined by EM.
May 2015
HaemMetabolome awarded to JJ Schuringa. Within this Marie Skłodowska-Curie Actions Innovative Training Network (H2020-ITN-EJD, 2.4M euro) we aim to unravel metabolomic changes in haematological malignancies. Besides the UMCG/Experimental Hematology, the consortium consists of Ulrich Gunther (University of Birmingham, UK; NMR-based metabolomics/fluxomics), Marta Cascante (University of Barcelona, Spain; LC-MS/MS based metabolomics) en Harold Schwalbe/Hubert Serve (University of Frankfurt, Germany; leukemias).
October 2014

Marta Capala published a paper in Plos ONE showing that Engulfment and Motility protein 1 (ELMO1) is upregulated in AML CD34+ stem/progenitor cells which predicts poor prognosis in normal karyotype AML.
October 2014

Dutch Cancer Foundation (KWF) basic science grant awarded to JJ Schuringa, Edo Vellenga and Vincent van den Boom, entitled: Functional characterization of the non-canonical PRC1.1 complex as novel target in leukemia. € 567.200,-
September 2014

Matthieu Bosman published a paper in Blood in which he identified TAK1 as a critical kinase that regulates leukemic stem cell self-renewal.
September 2014

Hein Schepers published his paper in Leukemia showing that CITED2 is critically important for leukemic stem cells.
June 2014

Publication of book chapter on ex vivo expansion protocols of human leukemic stem cells
March 2014

Bart Jan Wierenga published his paper in Plos ONE in which he studied hypoxia-induced transcriptome changes in human stem/progenitor cells and identified that TGFb and hypoxia pathways converge on cell cycle regulation.
January 2013

Vincent van den Boom published his study in Blood in which he showed that PRC1 members fulfil non-essential roles in human stem/progenitor cells.
May 2013

Francesco Bonardi published his study in Molecular and Cellular Proteomics. By performing genome-wide transcriptome and proteome studies he identified leukemia-specific plasma membrane proteins that can be used for identification and targeting of leukemic stem cells.
April 2013

Sarah Horton published her paper in Leukemia showing that fetal stem/progenitor cells readily transform along the myeloid of lymphoid lineage, depending on extrinsic cues, while adult human stem/progenitor cells can only be transformed along the myeloid lineage, in line whith what is seen in patients.
April 2013

JJ Schuringa co-authored a paper in Nature Reviews Cancer on cancer stem cells.
| Last modified: | 01 June 2021 10.25 a.m. |
