The evolution and suppression of male suicide under paternal genome elimination
Different genetic systems can be both the cause and the consequence of genetic conflict over the transmission of genes, obscuring their evolutionary origin. For instance, with paternal genome elimination (PGE), found in some insects and mites, both sexes develop from fertilized eggs, but in males the paternally derived chromosomes are either lost (embryonic PGE) or deactivated (germ line PGE) during embryogenesis and not transmitted to the next generation. Evolution of germ line PGE requires at least two transitions: (1) elimination of the paternal genome during spermatogenesis; (2) deactivation of the paternal genome early in development. So far, hypotheses for the evolution of PGE have mainly focused on the first transition. Interestingly, it has been shown in some scale insects that the second transition is triggered by maternal genes rather than the embryo’s own genes. We conjectured that this may have evolved as a response to suicidal tendencies of the embryo’s paternal genes and we developed models to investigate whether this argument is sound.
How could paternal genes be selected to favour their own destruction? Two observations are crucial to see this. First, in species with PGE paternal genes of males are never transmitted to the next generation – hence they have 'nothing to lose'. Second, paternal genes in males may gain inclusive fitness if the dead bodies of males can be 'recycled' to some extent and thereby improve their sisters’ survival prospects. Thus, in order for male suicide to evolve it seems necessary that there is some degree of sib competition over vital resources. Benjamin Normark noted that most taxa with PGE do indeed have strong levels of sib competition. However, he also observed that these species typically have high levels of sib mating and are therefore quite inbred. At first glance, one might expect inbreeding to counteract the spread of paternal male suicide as it can lead to increased relatedness between the maternal and paternal genome of an individual. However, inbreeding also increases relatedness between sibs, which might promote male suicide. To make matters even more complicated, a life history with inbreeding and sib competition tends to induce selection for female-biased sex ratios, thus increasing the reproductive value of individual males, which might be an additional obstacle to the evolution of male suicide. Clearly, a formal model is required to investigate the balance of these opposing effects.
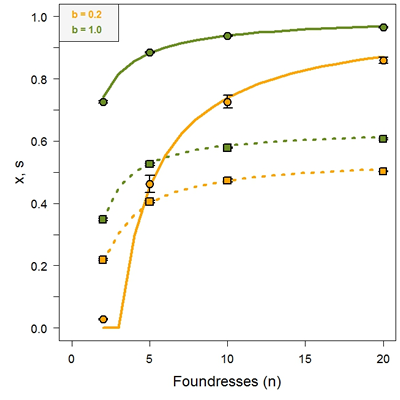
In our model (Ross, Shuker & Pen, Evolution, 2010), we considered the fate of a partially suicidal gene that is expressed in males by the paternally inherited half of their genome. Specifically, the gene causes a probability x of suicide, and we let the value of x evolve. A parameter b was introduced as a measure of the recycling efficiency (between 0 and 1) of dead males. We allowed for some degree of inbreeding by assuming that the population is subdivided in standard-sized patches of n mated females whose offspring mate randomly on their natal patch followed by dispersal of newly mated females. These ingredients were incorporated in an analytical kin selection model to study what level of suicide, if any at all, is expected to evolve depending on recycling efficiency (as determined by b) and the level of inbreeding (as determined by n).
Some results are shown in Figure 1. Male suicide can evolve, and unless some inbreeding occurs, 100% suicide evolves and population extinction will be the result, regardless of the efficiency of recycling. If maternally-controlled sex ratios are allowed to co-evolve, male-biased primary sex ratios are possible, in contrast to standard models that predict female-biased sex ratios for populations with inbreeding. Maternal suppression of male suicide is always selected for, leading to complete suppression of male suicide, even if dead males are recycled with 100% efficiency.
| Last modified: | 03 December 2015 12.54 p.m. |
